Signatera™ Genome: the only genome-designed MRD assay with Medicare Coverage
Learn MoreSignatera™ Genome: the only genome-designed MRD assay with Medicare Coverage Learn More
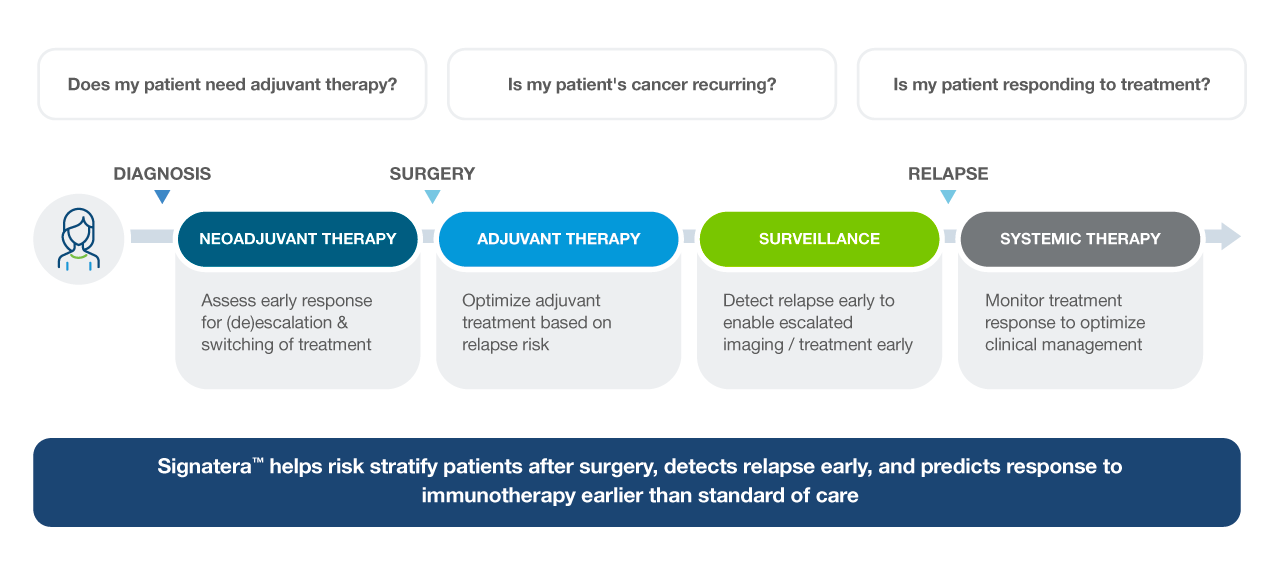
Signatera™ Indications
Signatera™ can be used for a variety of cancer types. To learn more about how, please select one.
See how Signatera™ analyzes circulating tumor DNA (ctDNA) to assess molecular residual disease (MRD)
- ctDNA is a powerful tool that can be measured to assess the absence or presence of MRD.
- Signatera™ is a highly sensitive and personalized MRD assay using ctDNA and is custom designed for each patient to help identify relapse earlier than standard of care tools.
Signatera™ is the most extensively validated and widely used MRD assay available¹
Broadest patient access. Covered by Medicare for monitoring disease progression, disease recurrence, or relapse for patients with multiple solid tumors
Experience
50%
US clinicians ordered.
Validation
189
peer reviewed publications.
Powering treatment decisions from diagnosis through survivorship
How Signatera™ Works: a personalized and tumor informed approach to MRD detection

Personalized, tumor-informed assay
One-time, primary tissue sample and matched normal sample is required for whole exome or whole genome sequencing and personalized test design.
Ultrasensitive ctDNA detection
Signatera™ is designed to detect ctDNA of somatic and truncal variants to optimize sensitivity. Tumor-informed methid enables filtering of CHIP mutations to decrease false positive rates.
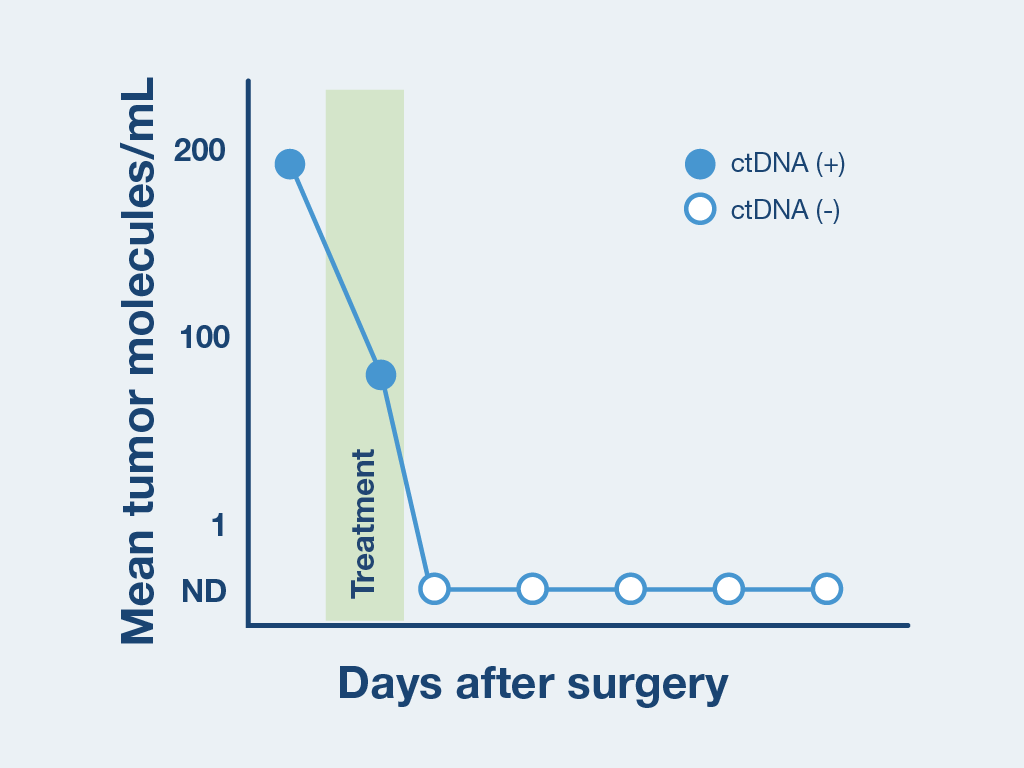
Optimized for longitudinal monitoring
Once the patient’s personalized test has been designed, only a blood sample is needed each subsequent time.
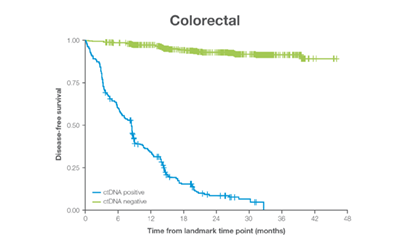
In clinical studies, Signateraᵀᴹ showed high performance across multiple cancer types
Complementing current tools, Signatera™ provides clinically meaningful MRD assessment to stratify patients after surgery or treatment.
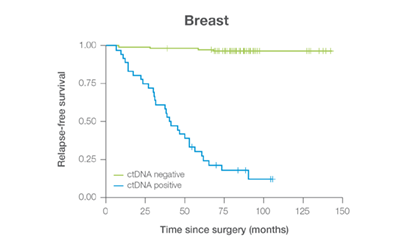
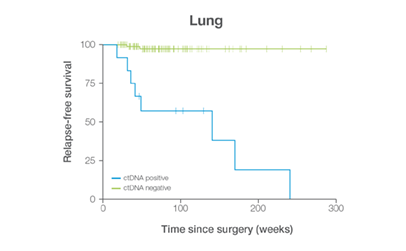
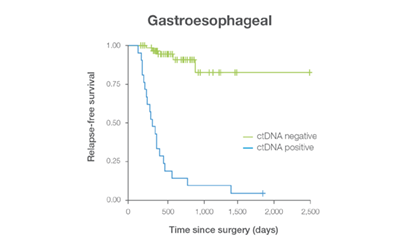
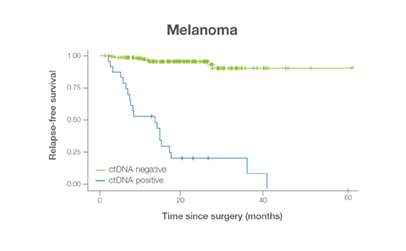
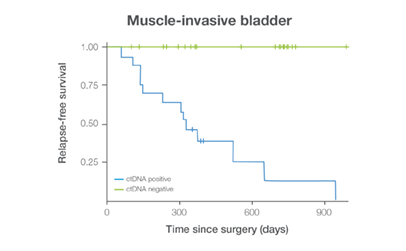
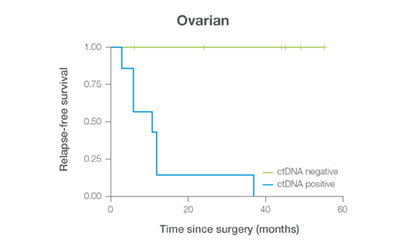
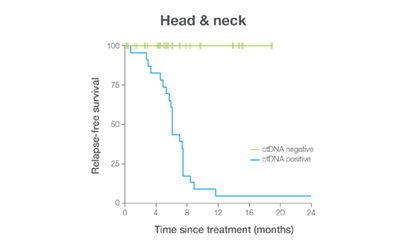
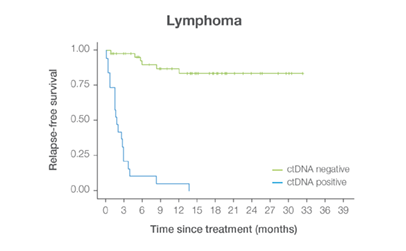
Signatera™ Genome sequences deeper on the most critical variants to provide a more clinically meaningful MRD signal
- Ultra-deep sequencing (up to 350,000x coverage for each selected variant) DRFS by ctDNA Status¹
- Highly curated selection of 64 clonal variants for optimized balance of ultrasensitivity and high specificity²
- Proprietary variant selection and calling algorithms for bespoke MRD monitoring

The most widely trusted MRD assay available
Established experience
Signatera™ has been ordered by >50% of US oncologists and ordered for more than 400,000 patients.1,2
Reliable service and support
- 90% of initial Signatera™ reports are provided within 3 weeks and 90% of subsequent reports are provided within 1 week.1,2
- Natera offers complimentary mobile phlebotomy, genetic information sessions and Patient Portal access to Signatera™ patients.
Broadest patient access
Signatera™ is covered by Medicare for monitoring disease progression, disease recurrence and relapse.

Having the information about what is happening in our bodies, in our blood, with our cancer, gives us the ability to be proactive and to take charge …and feel like we are in a position of strength. And I am thankful for that.
Meet Amanda, Colorectal Cancer Survivor and Signateraᵀᴹ Patient
Amanda shares her powerful and unexpected where a routine genetic screening during her pregnancy led to a Stage IV colon cancer diagnosis. Watch Amanda discuss her experience as a survivor, balancing life as a new mom, and why Signateraᵀᴹ is vital to her life after cancer treatment.
Is Signatera™ right for your patients?
We’re here to help you find out
1George M, et al. Poster presented at the 2025 ASCO Annual Meeting, Chicago, IL, June 2025.
2Natera Data on File as of May 1st, 2026.