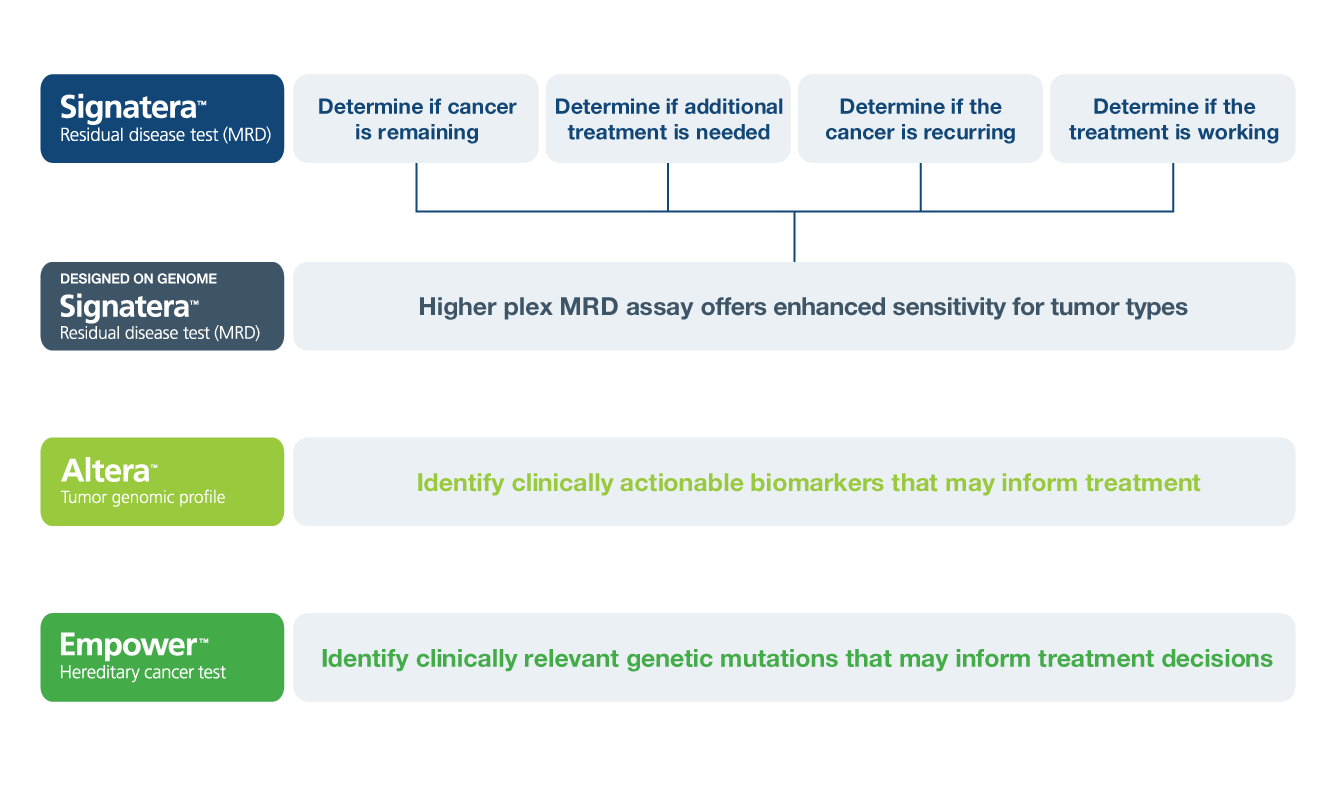
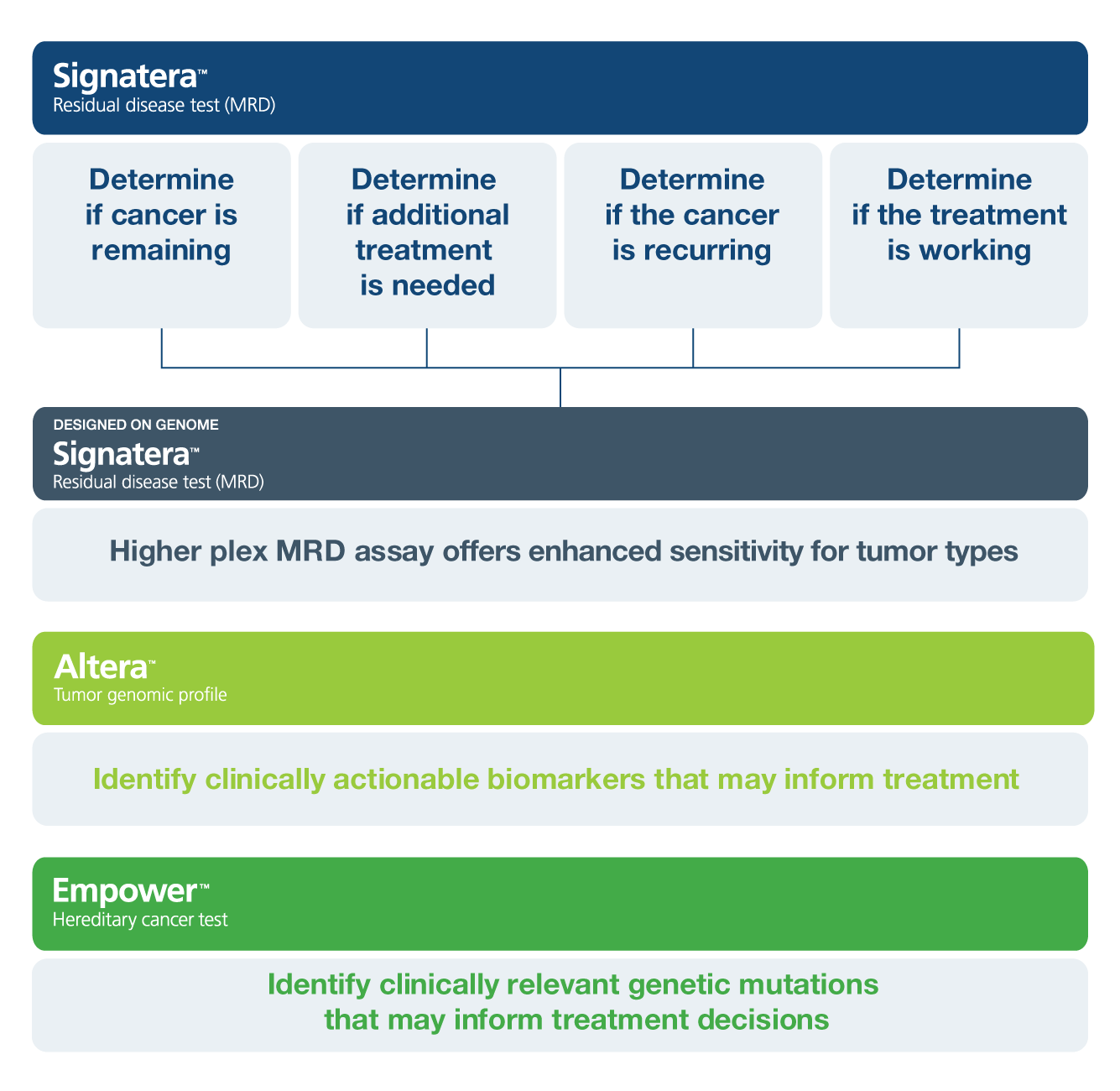
Inform Decisions for High Risk Patients
Risk-Stratify Patients
Identify patients with residual disease after surgery who may benefit from adjuvant treatment for high risk individuals
5.6x
higher risk of distant metastasis in patients with molecular residual disease (MRD) after surgery1
Detect Recurrence Earlier
Use Signatera™ serially to monitor and identify breast cancer patients at high risk of recurrence2
52x
higher risk of relapse in ctDNA positive versus ctDNA negative patients
Assess Treatment Response
Track changes in ctDNA levels to evaluate treatment response3
6 weeks
to understand response to immunotherapy treatment3
Signatera™ Genome demonstrated ultrasensitive MRD detection down to 1 part per million (PPM)11
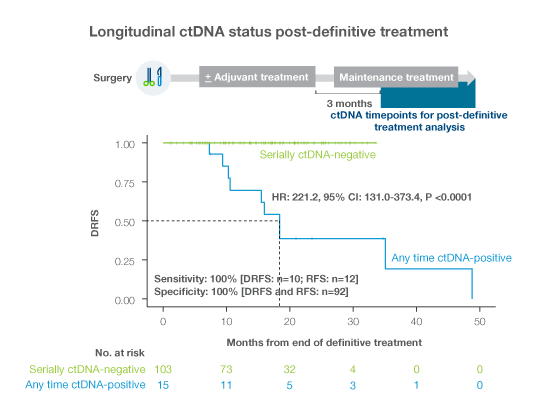
Signatera™ was shown to be highly prognostic of patient outcomes and predictive of adjuvant treatment benefit.
Signatera™ Genome: Evolving MRD from the most trusted assay in breast cancer
As Natera continues to lead the way in precision oncology, Signatera™ Genome has demonstrated exceptional performance in breast cancer, offering unparalleled sensitivity and specificity for detecting residual disease and informing critical treatment decisions.
Get Started
Detect Recurrence Earlier
- Up to 30% of breast cancer patients with no evidence of disease after curative intent treatment will eventually relapse and succumb to their disease4 — suggesting that existing tools are inadequate at identifying recurrence before the patient becomes symptomatic
- Approximately 10-15% of patients experience reduced performance status upon cancer recurrenceand treatment options are limited5
- Longitudinal testing with Signatera™ has been shown to identify patients at high risk of recurrence with 88% sensitivity and a median of 10.5 months (range 0-38 months) ahead of imaging2, potentially enabling meaningful and timey intervention
Inform Questions Across Breast Cancer Treatment
Explore the Data in Breast Cancer
Neoadjuvant Response Monitoring and Risk Stratification
I-SPY 2 Study Schema
I-SPY 2 Studies: Can ctDNA Refine pCR as a Surrogate Endpoint for Survival?
For patients treated with neoadjuvant therapy, achieving a pathologic complete response (pCR) generally denotes a favorable prognosis. However, how can providers risk-stratify patients who have residual disease?
I-SPY 2 ctDNA Objectives
- Evaluate ctDNA as a biomarker for monitoring response to neoadjuvant therapy6,7
- Assess prognostic value of ctDNA in stratifying patients for risk of early metastatic recurrence6,7
The I-SPY 2 Platform Master Protocol
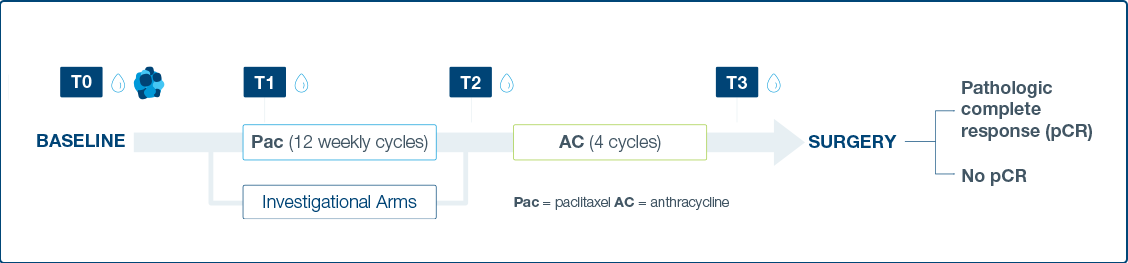
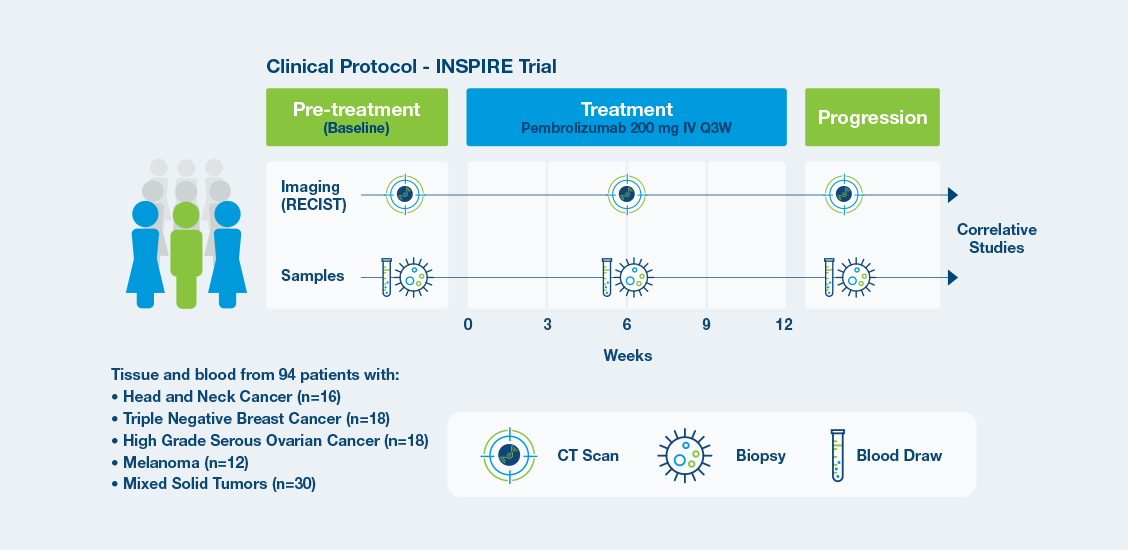
- Response to neoadjuvant chemotherapy (NAC) and investigational treatments assessed through ctDNA dynamics at four time points
- ctDNA derived from plasma was assessed at four time points: pretreatment (T0), 3 weeks after paclitaxel/study drug initiation (T1), between
paclitaxel/study drug and anthracycline regimens (T2), and prior to surgery (T3).
I-SPY 2 Results
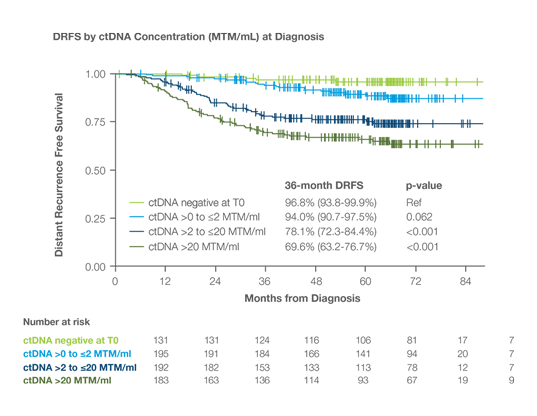
ctDNA concentration (MTM/ml) at diagnosis correlated with higher risk recurrence, regardless of receptor subtype
Study Cohort: 712 high-risk early-stage bre with HR+/HER2-, TNBC, HER2+ disease1
MRD testing and impact on DRFS: Patients ctDNA prior to NAT had 96.8% 3-year DRFS
pCR and ctDNA status Can Help Risk-Stratify Patients6,7
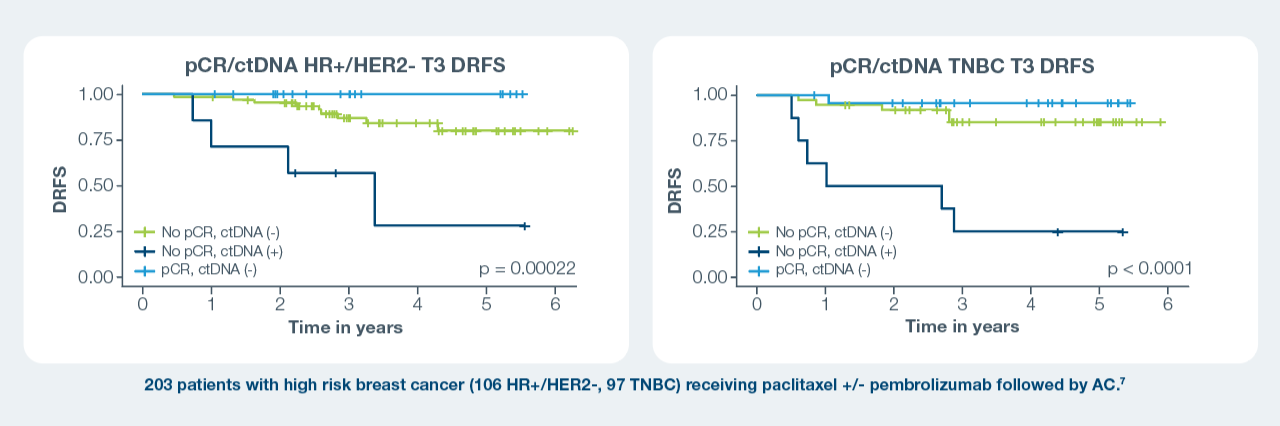
At T3 across subtypes:
- Patients who achieved a pCR and cleared their ctDNA had the best survival outcomes; those who did not achieve a pCR and were ctDNA positive
had the worst outcomes.1–3 - Regardless of pCR status, persistent ctDNA positivity by T3 was associated with shorter DRFS compared to ctDNA negativity.1–3
Adjuvant MRD Assessment
Risk stratify patients after surgery to help identify those with residual disease who may benefit from intensified or extended adjuvant treatment
The EBLIS Study:
ctDNA status in conjunction with existing tools after surgery and adjuvant chemotherapy identified high risk patients with poor recurrence-free survival8 who may benefit from adjuvant treatment for high risk individuals
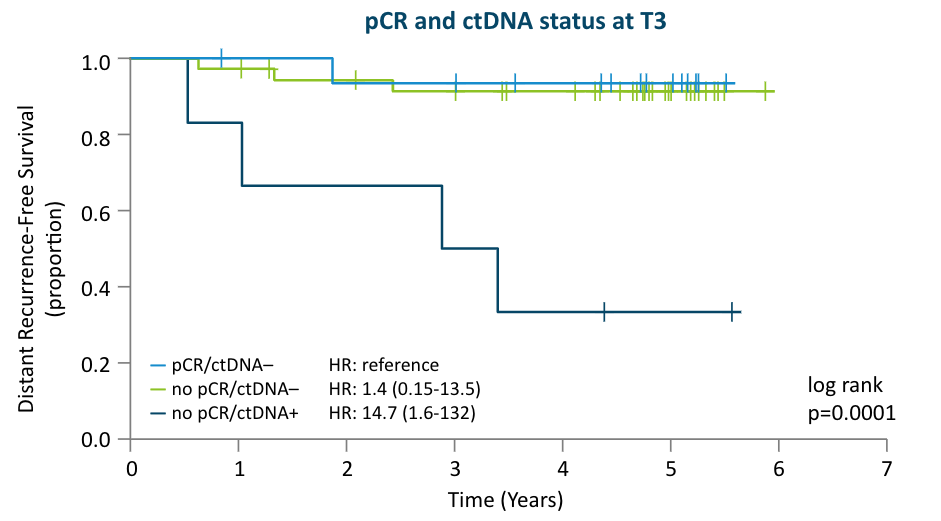
The I-SPY 2 Study:
Prior to surgery, the I-SPY 2 study showed that lack of ctDNA clearance regardless of pCR status identified patients with poor response to neoadjuvant chemotherapy and a high risk of recurrence6, which can help inform adjuvant treatment decisions
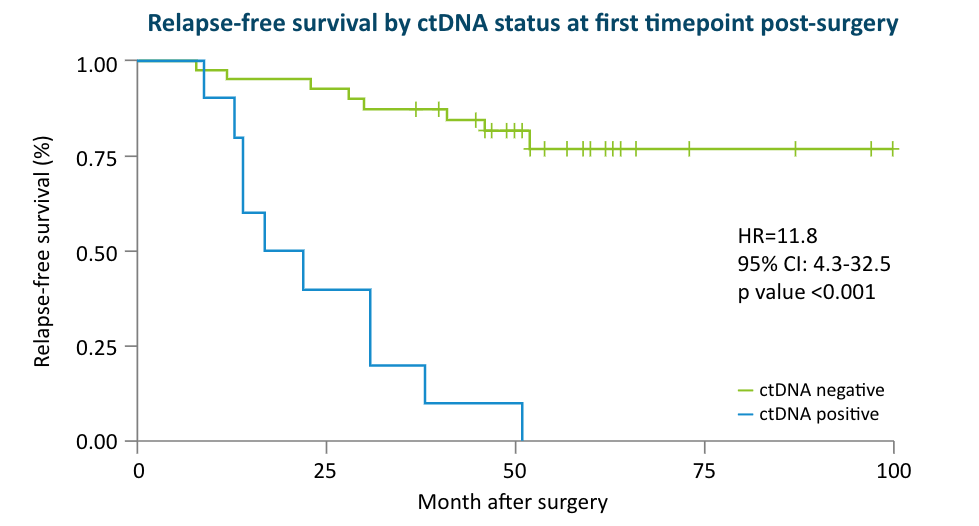
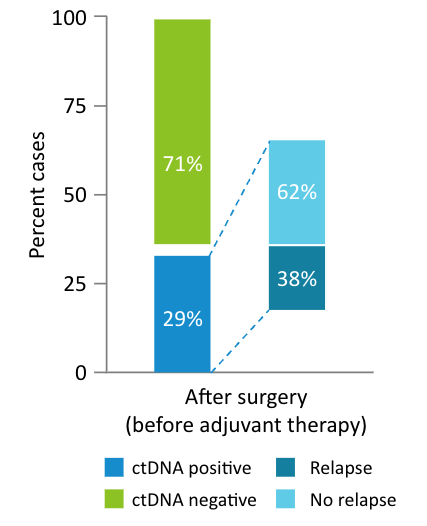
The Early Breast Cancer Study:
48 early stage breast cancer patients receiving primary surgery included in analysis (40 also received chemotherapy), with 144 total plasma samples; median follow up time 5 years (0.5-6)1

- 38% of patients who were ctDNA positive after surgery relapsed, while the remaining possibly benefited from adjuvant therapy.1
- Detection of ctDNA after surgery was associated with worse outcomes compared to no ctDNA detection (DMFS HR: 5.6; 95% CI: 1.1-29.3; P=0.04)1
- These data suggest that although MRD positivity post-surgery is prognostic of patient outcomes, it may be modifiable with adjuvant therapy.1
Surveillance Recurrence Monitoring
Dare trial: Provided insights into the role of ctDNA to guide adjuvant therapy decisions in high-risk eBC
- 12% of patients were found to be ctDNA+ during surveillance9
- 73% of ctDNA+ patients were imaging negative for metastatic disease (i.e. had molecular relapse)9
- 93% of patients with molecular relapse were willing to be randomized9
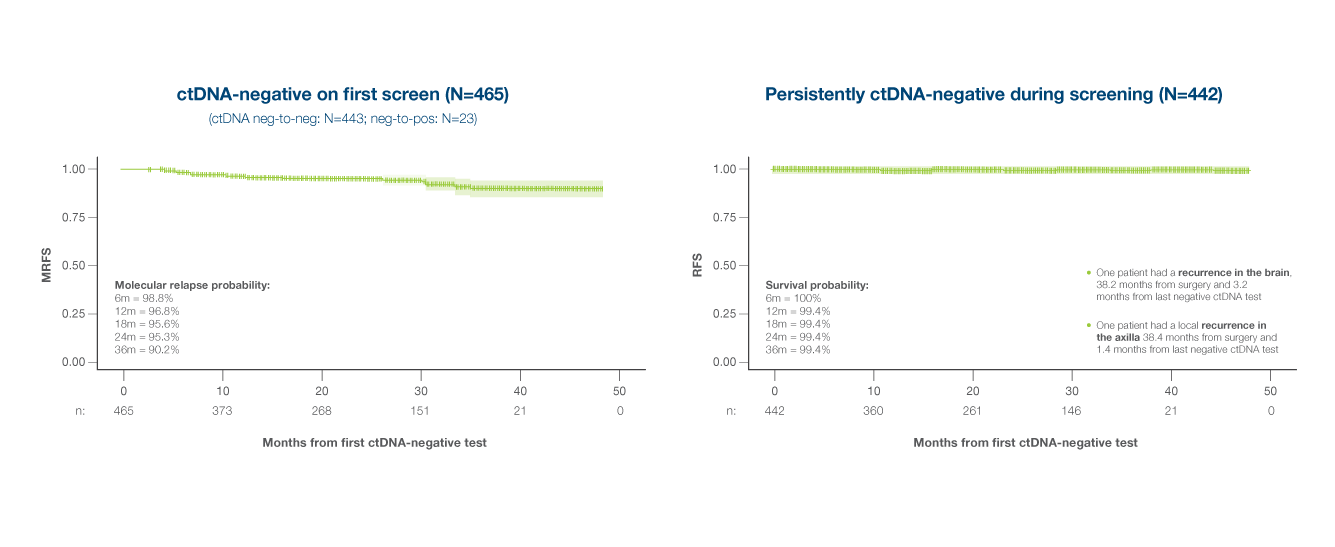
- ctDNA negativity was strongly associated with improved RFS, with a NPV of 99% with a median follow-up of 27.4 months9
Association of early on-treatment ctDNA dynamics with RFS
- 1 of 9 patients with ctDNA clearance or decrease at 3 months post randomization had clinical relapse
- 6 of 10 patients with increasing ctDNA levels had clinical relapse
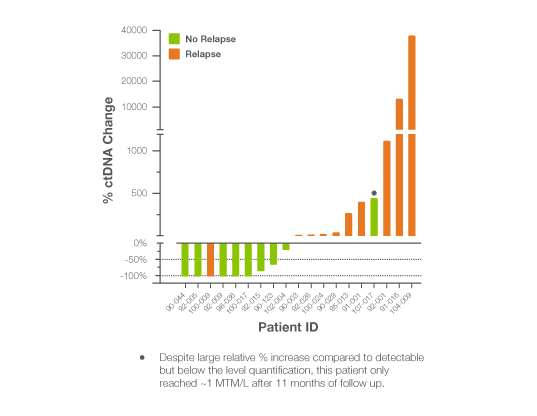
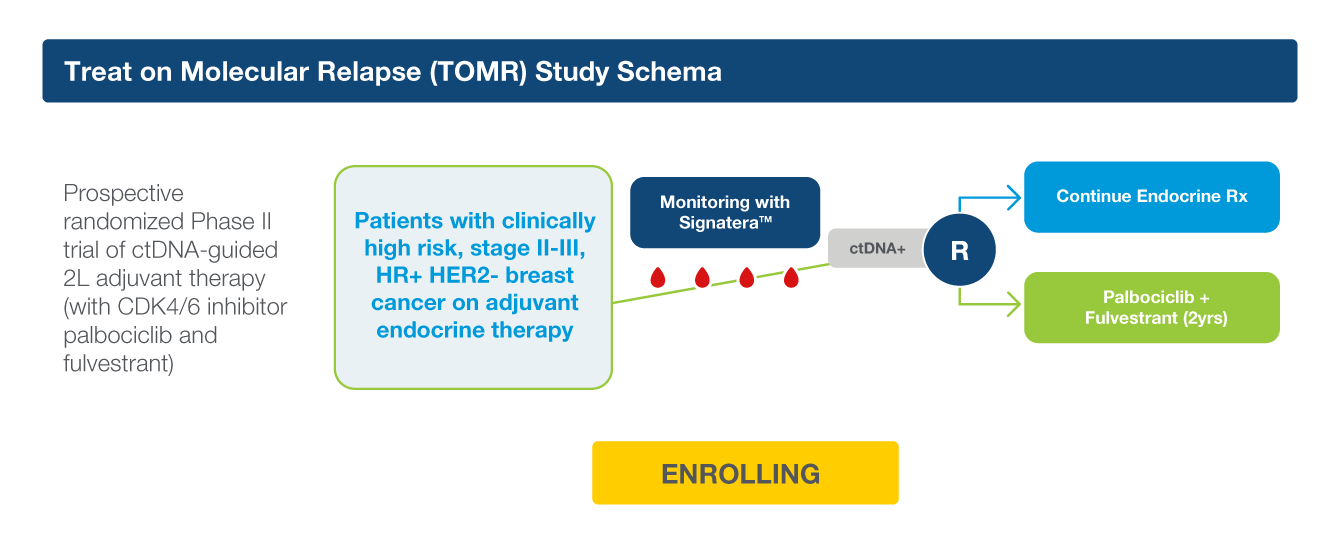
The EBLIS Study: Is there a more sensitive technology that can detect recurrence earlier?
The Exploratory Breast Lead Interval Study (EBLIS) explored the potential use of ctDNA monitoring in patients with high risk breast cancer in the longest ctDNA follow up study to date.2
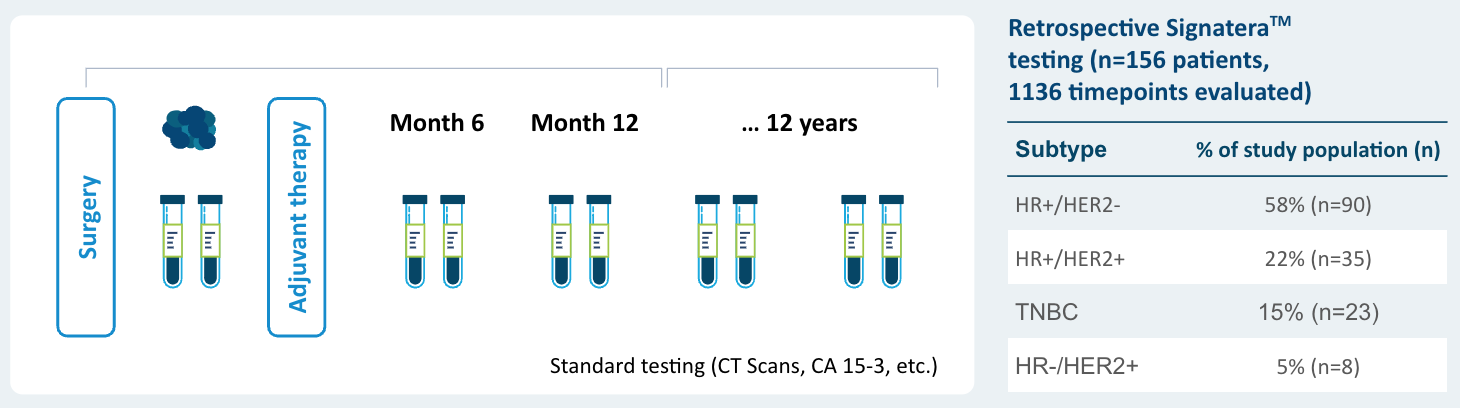
EBLIS Protocol
EBLIS Results
Signatera™ can be used serially to monitor and identify breast cancer patients at high risk of recurrence
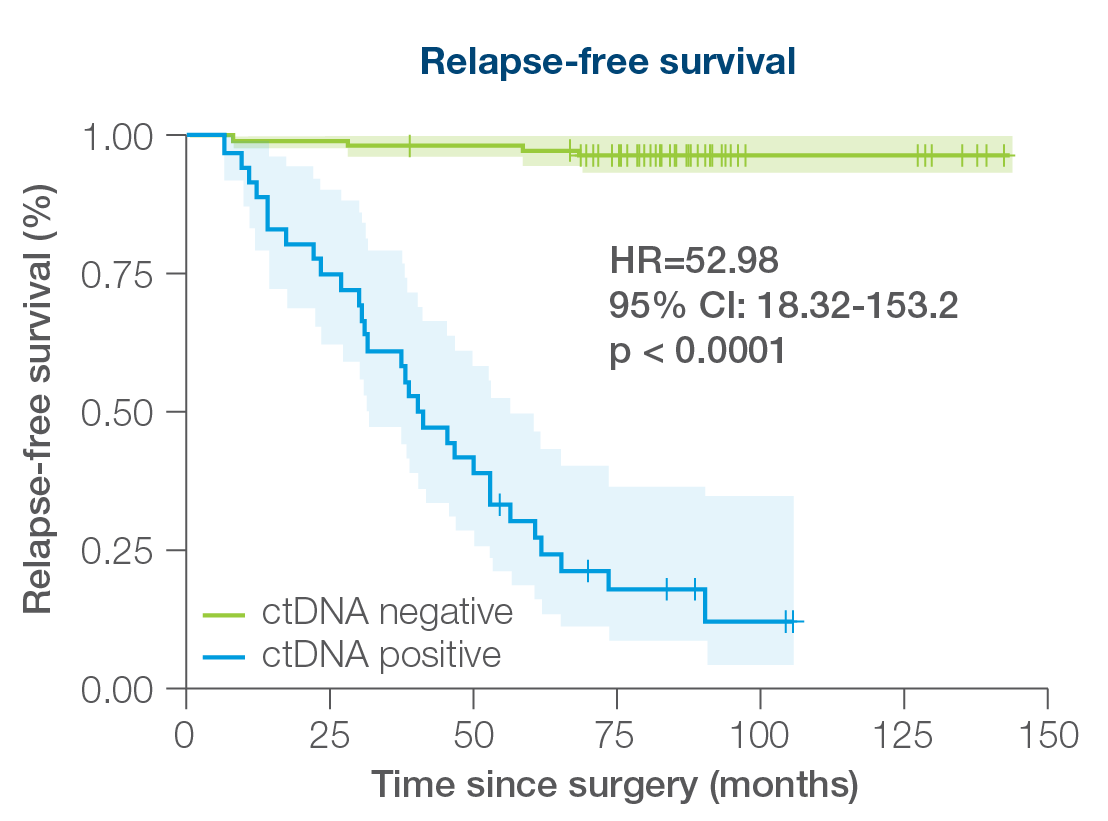
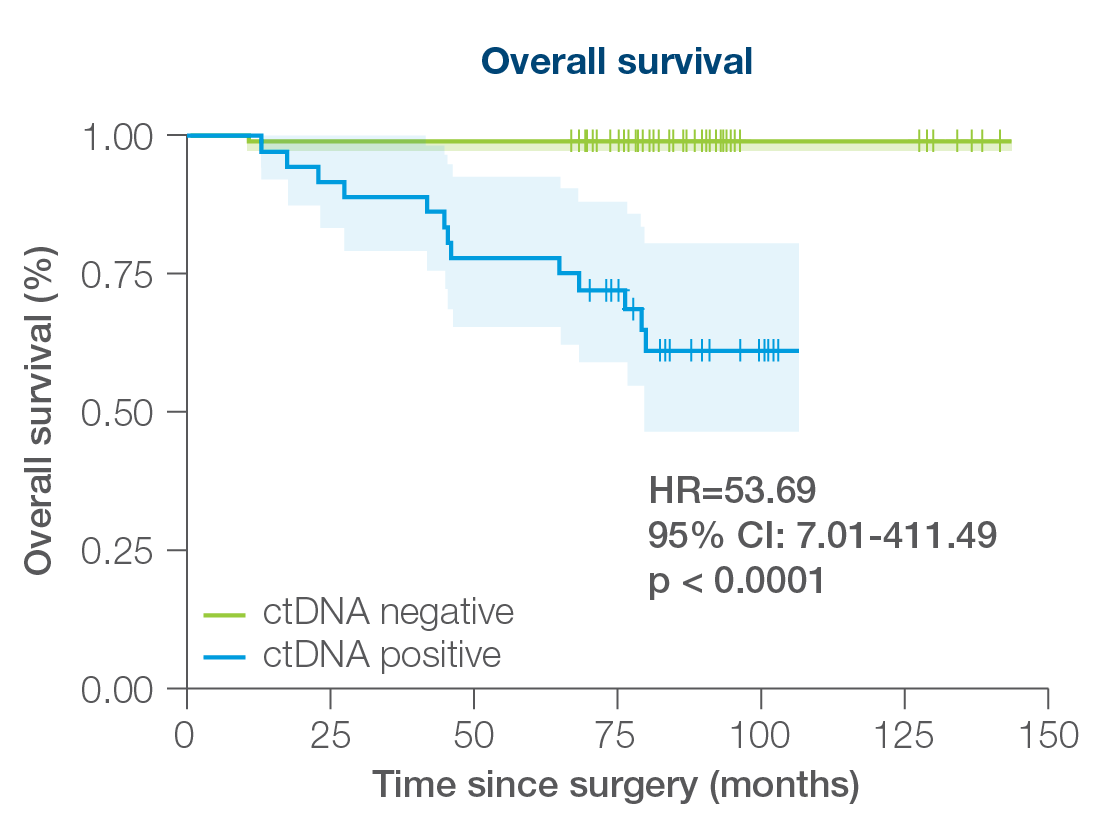
- 156 patients followed over a peroid of 12 years including all breast cancer subtypes
- 88% sensitivity: Signatera™ detected ctDNA in 30 out of 34 patients before clinical or radiologic relapse
- 10.5 months median lead time over radiographic recurrence (range 0-38 months)
In the largest ctDNA analysis of HR+/HER2- lobular breast cancer to date (n=7,381), ctDNA-positivity was linked with higher risk of recurrence and earlier relapse.1
Data highlights: Signatera™ brings clarity to lobular breast cancer risk.
- 7,381 Patients: This is the largest real-world study of lobular breast cancer evaluating ctDNA outcomes.
- Early recurrence: Within two years of surgery, ctDNA-positivity identified aggressive disease across subtypes, including HR+/HER2- lobular breast cancer.
- After 2 years: ctDNA-positivity continued to identify patients at risk for late relapse – a hallmark of lobular breast cancer.
Assess Immunotherapy Response
The INSPIRE Study: Can ctDNA be Validated as an Early Biomarker of ICI Treatment Response?
Despite dramatic improvements in cancer care using antibodies that block immune checkpoint proteins, less than 20% of eligible patients will derive sustained response or clinical benefit to immune checkpoint inhibitors (ICIs).7 How can ctDNA accurately identify those patients who are benefiting from ICI treatment?
INSPIRE Objectives
The prospective phase II INSPIRE trial addressed clinically relevant issues related to monitoring response to ICIs by assessing baseline ctDNA status and ctDNA dynamics.
INSPIRE Protocol
INSPIRE Results
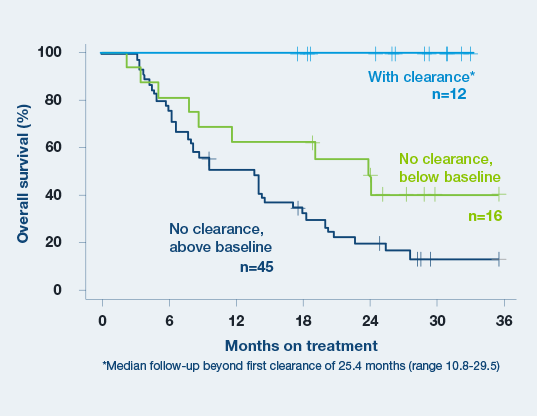
Signatera™ predicts immunotherapy benefit as early as week 6.3
Baseline ctDNA concentration and ctDNA dynamics during treatment correlated with progression-free survival, overall survival, clinical response, and clinical benefit.3
- A decrease in ctDNA levels from baseline to the beginning of cycle 3 is a strong predictor of increased OS and PFS.3
- Clearance of ctDNA at any time point resulted in superior clinical outcomes. All 12 patients who cleared ctDNA for at least one on-treatment time point remained alive during follow-up period (median follow-up beyond first clearance of 25.4 months)3
Monitor Metastatic Treatment
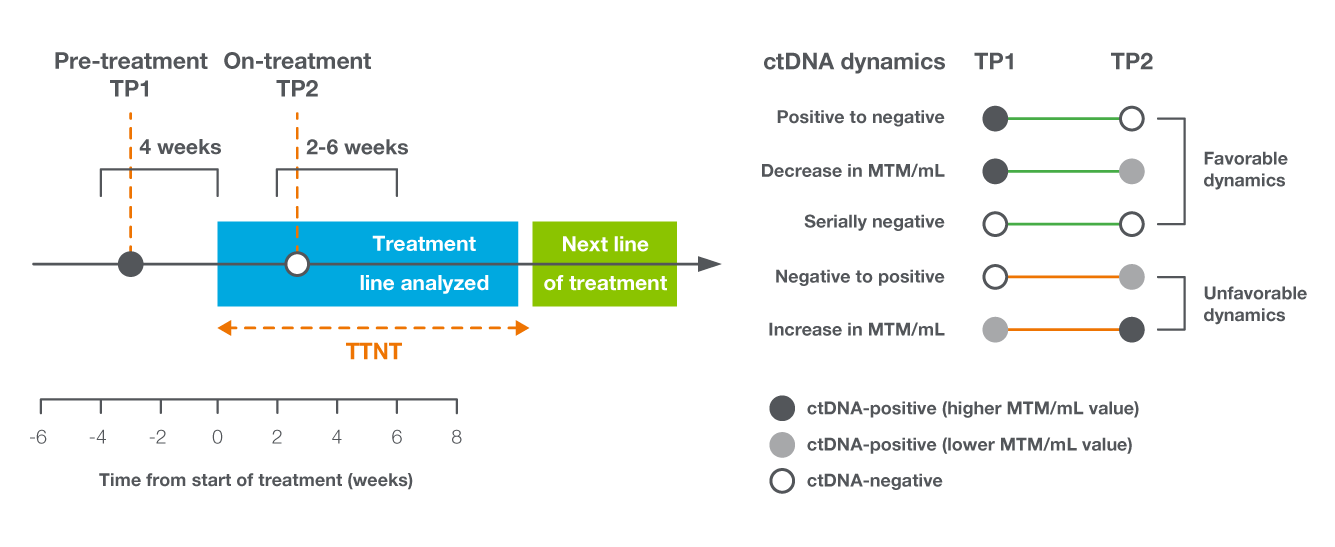
In the metastatic setting, ctDNA dynamics served as a predictor of treatment response in mBC
- Study objective: To determine the relationship between early on-treatment ctDNA dynamics and real-world treatment outcomes10
- Utilized real-world database linked to claims data to evaluate the association between ctDNA dynamics and the time to next treatment (TTNT) in patients with 616 mBC10
Refined risk stratification in eBC HR+/HER2–
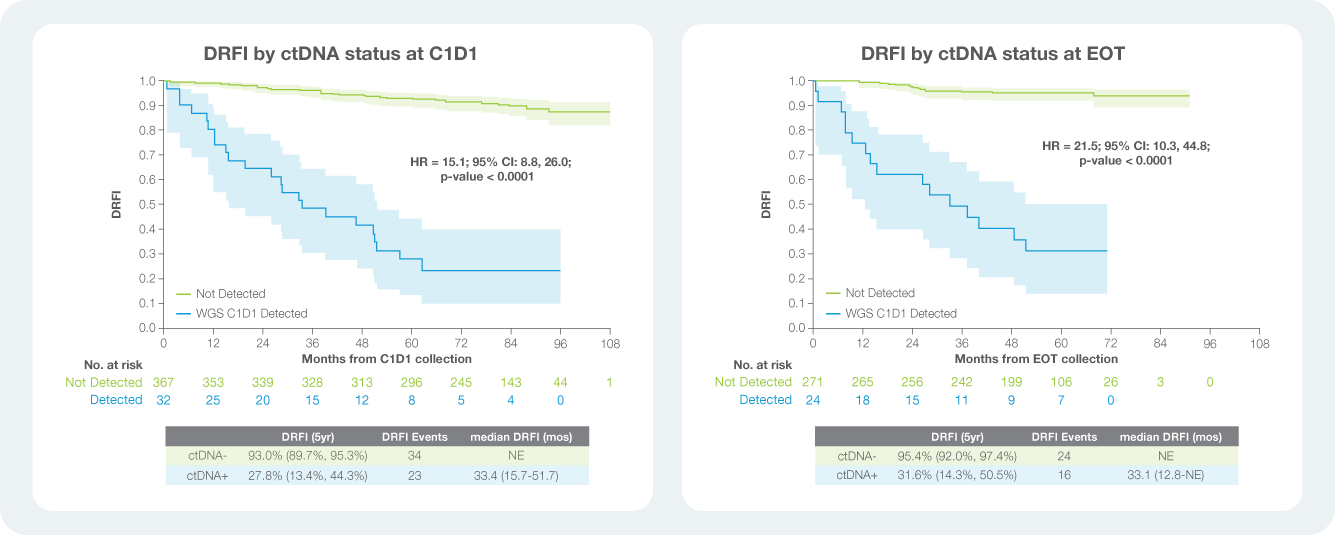
Signatera™ genome negativity identifies a large proportion of the study population with exceptionally low recurrence risk.
The PALLAS Study:
- Signatera™-positivity at C1D1 was associated with an annualized distant recurrence risk of about 15%, while Signatera™-negativity was associated with an annualized risk of about 1.4%
- Risk stratification by Signatera™ status remained durable over time, distinguishing a high-risk subgroup with baseline positivity despite overall low positivity in HR+/HER2– disease
- Most patients initiated endocrine therapy prior to the C1D1 blood draw, with approximately 80% starting treatment before collection
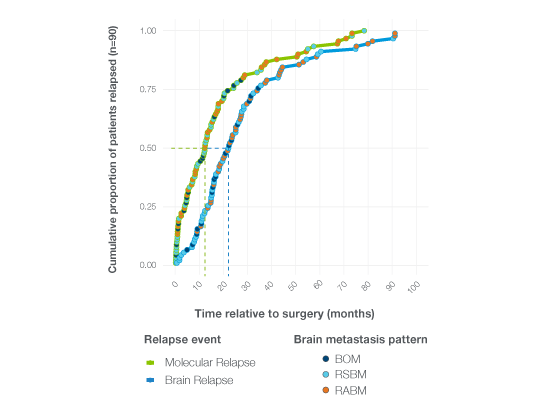
Early Detection of Brain Metastases in Breast Cancer Using ctDNA Surveillance
- 83% of patients who developed brain metastases had detectable ctDNA prior to clinical relapse
- 62% of patients with brain-only metastases were ctDNA-positive before diagnosis
- Median 10-month lead time from ctDNA detection to clinically confirmed brain metastasis
- Lower ctDNA levels were observed in brain-only relapse compared to extracranial metastatic disease, highlighting biologic differences in tumor shedding
Surveillance to Detect Breast Cancer Recurrence, Including Brain Metastases
- Positive ctDNA results frequently preceded radiographic or symptomatic brain metastasis, even when the brain was the first site of recurrence
- Detection of ctDNA prompted follow-up imaging in real-world practice, supporting its role as an early risk signal during surveillance
- Findings suggest ctDNA surveillance may help identify patients who could benefit from earlier CNS evaluation
Maximize Patient Care: Identify More Eligible Breast Cancer Patients for Hereditary Cancer Testing
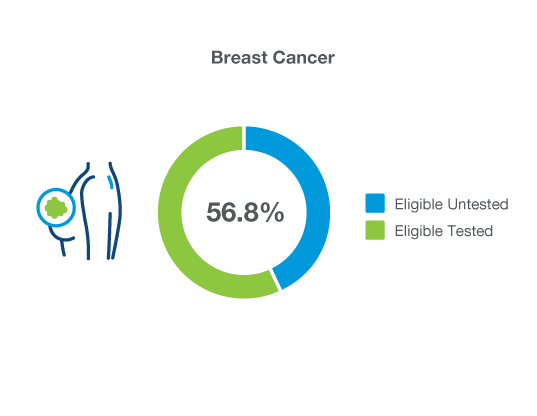
Breast cancer patients ≤50 years of age and patients with TNBC are eligible for hereditary cancer testing, however just 56.8% are actually receiving it

A pan-cancer, multi-gene assay designed to identify germline mutations that may inform cancer treatment options, risk for secondary cancers, and hereditary risk information for family members.
Oncology Solutions
Covered by Medicare for multiple solid tumor indications
Learn More
Is Signatera™ for breast cancer right for your patients?
References
1Cutts R, et al. Detection of ctDNA following surgery predicts relapse in breast cancer patients receiving primary surgery. SABCS; December 7-11, 2021.
2Shaw JA, et al. Serial postoperative ctDNA monitoring for early detection of breast cancer recurrence. ASCO; June 3-7, 2022.
3Bratman SV, et al. Nature Cancer. 2020;1(9):873-881.
4Early Breast Cancer Trialists’ Collaborative Group. Lancet. 2005;365:1687-1717.
5O'Connor T, et al. Clin Adv Hematol Oncol. 2013;11(6):341-347.
6Magbanua MJM, et al. Ann Oncol. 2021; 32(2):229-239.
7Magbanua MJM, et al. Comparison of the predictive and prognostic significance of circulating tumor DNA in patients with high risk HER2-negative breast cancer receiving neoadjuvant chemotherapy. AACR; April 8-13, 2022.
8Coombes RC, et al. Clin Cancer Res. 2019 Jul 15;25(14):4255-4263.
9Lajos Pusztai, et al. Circulating tumor (ct)DNA monitoring of ER+/HER2- high-risk breast cancer during adjuvant endocrine therapy. Presented at the American Society Clinical Oncology annual meeting; 2025; Abstract 1010.
10Pedram Razavi, et al. Circulating tumor DNA (ctDNA) dynamics as a predictor of treatment response in metastatic breast cancer (mBC). Presented at the American Society Clinical Oncology annual meeting; 2025; Abstract 1011.
11George et al. (2025, May 30–June 3). Clinical performance of Signatera Genome assay in a cohort of patients (pts) with solid tumors [Poster presentation]. 2025 ASCO Annual Meeting, Chicago, IL. Abstract #3142.










