Vasistera™
Limited noninvasive prenatal testing (NIPT)
Vasistera™ NIPT is a blood-based genetic, prenatal screening test of the pregnant person that screens for common chromosomal conditions that affect a baby’s health. Vasistera™ screens for trisomy 21, trisomy 18, trisomy 13, and sex chromosome aneuploidies. Fetal sex reporting is optional. Vasistera™ can be performed as early as ten weeks gestation. Most results will be returned to your doctor within 5-7 calendar days.
Like Panorama™, our company’s market-leading NIPT, Vasistera™ NIPT leverages Natera’s unique SNP*-based methodology to analyze cell-free DNA. Unlike Panorama™, Vasistera™ NIPT does not report other chromosomal conditions, such as triploidy, or microdeletions such as 22q11.2 deletion syndrome.
Vasistera™ NIPT is a screening test, which means that this test does not make a final diagnosis. A screen positive result means that your pregnancy has a higher chance of having a specific genetic condition. However, you cannot know for sure if your baby has that condition based upon the screening result alone. All medical decisions should be made after discussion with your clinician regarding diagnostic testing during the pregnancy, like chorionic villus sampling (CVS) or amniocentesis, or testing the baby after birth.
*SNP, single nucleotide polymorphism

Find out how Natera’s prenatal screening tests, including Vasistera™, helps identify your baby’s risk of having a genetic condition
Conditions Screened For
Vasistera™ NIPT screens for common genetic conditions that are caused by extra or missing chromosomes in the baby’s DNA. Because Vasistera™ NIPT uses a unique technology to distinguish between the pregnant person’s and the baby’s DNA, it can avoid known sources of error that affect other NIPTs.1 The conditions screened, such as Down syndrome, are caused by extra copies of a specific chromosome.1
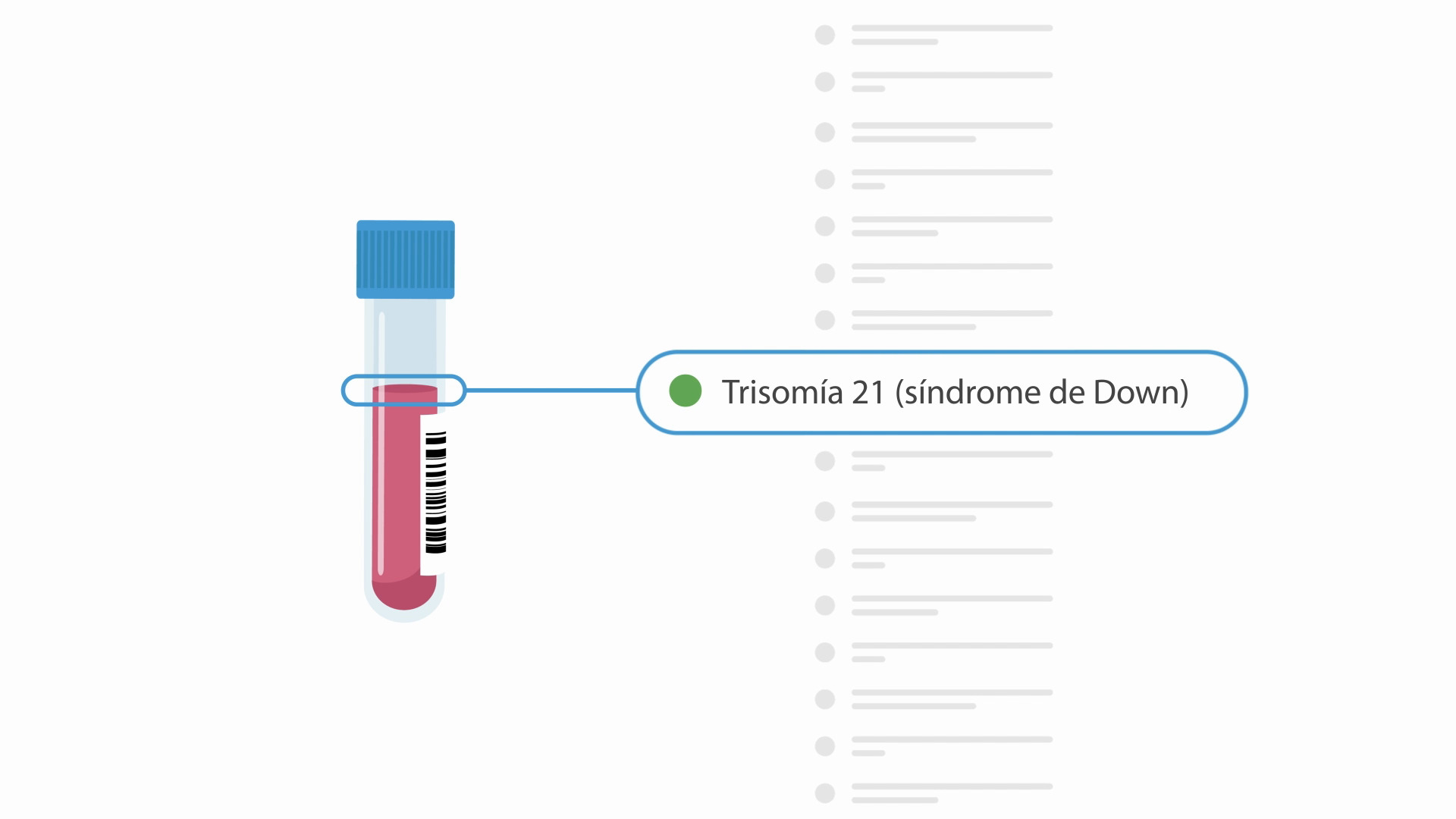
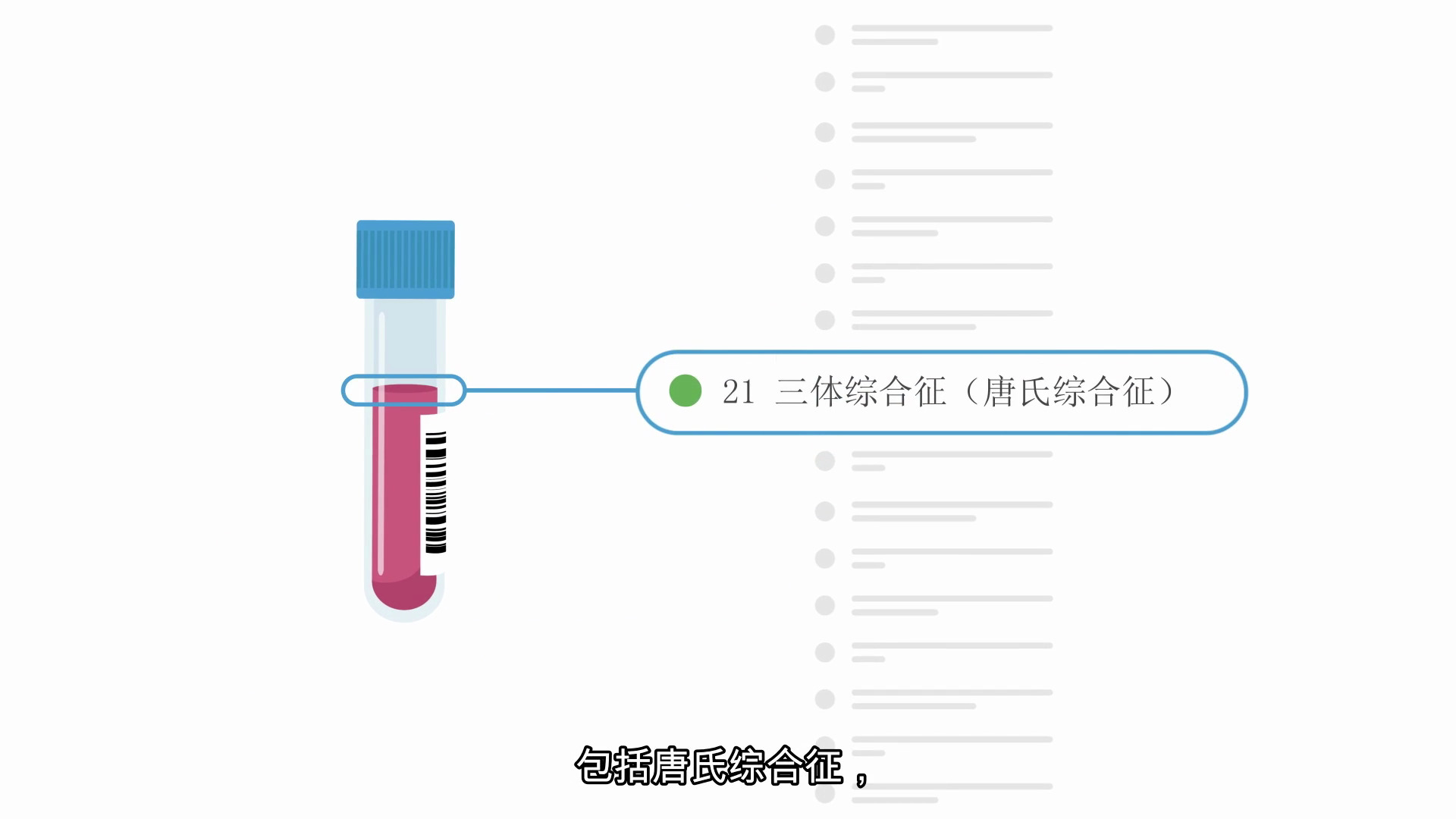
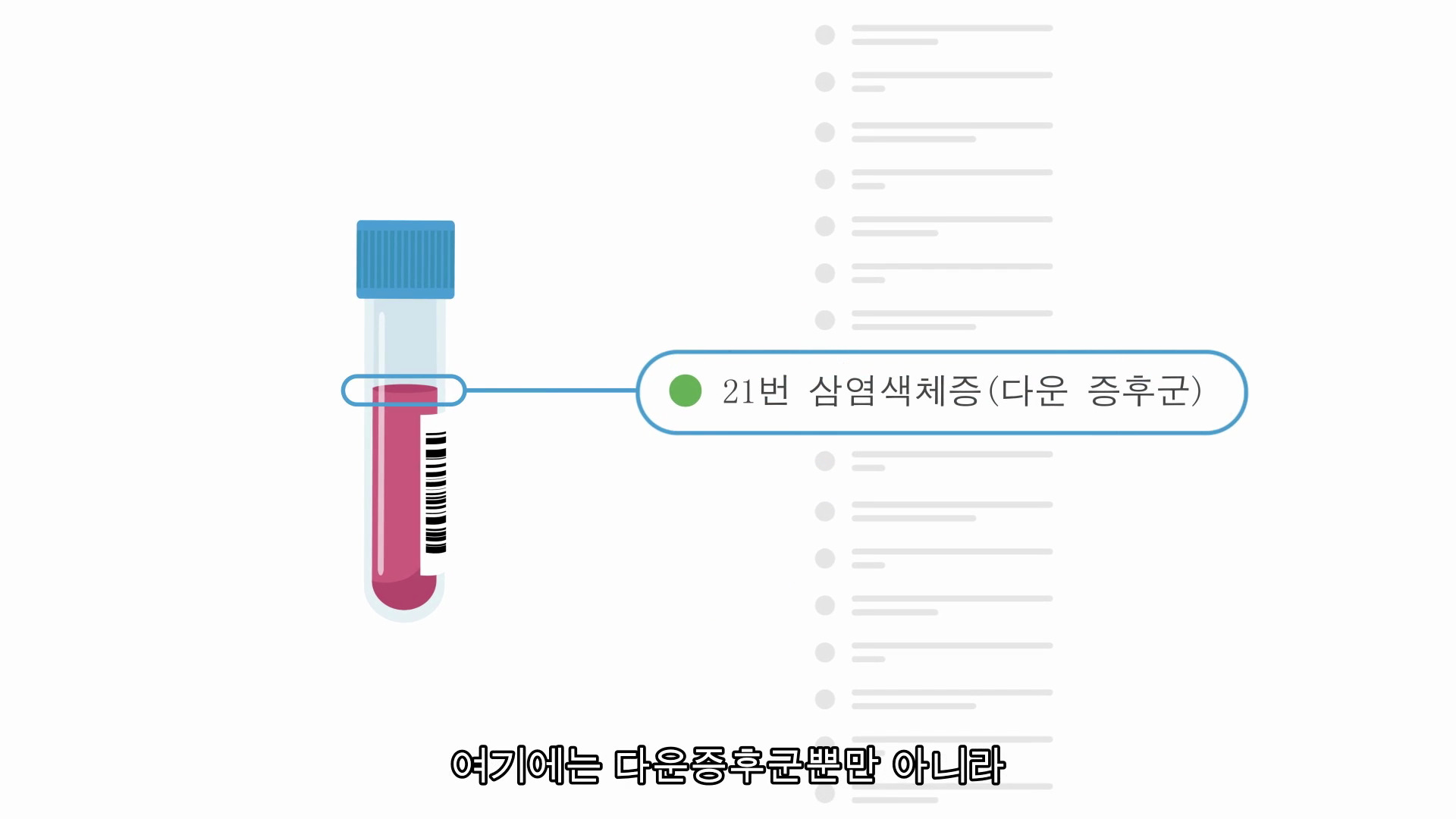
TRISOMIES*
Trisomy 21 (Down syndrome)
Babies with Down syndrome have three copies of chromosome 21 and have intellectual disabilities that range from mild to severe. Children with Down syndrome will need extra medical care depending on the child’s specific health problems. Early intervention has allowed many individuals with Down syndrome to lead healthy and productive lives. The presence of medical conditions, like heart defects, can affect the lifespan in these children and adults; however, most individuals with Down syndrome will live into their 60s. Miscarriage occurs in about 30% of pregnancies with Down syndrome while overall about 1 in 700 babies are born with Down syndrome.
Trisomy 18 (Edwards syndrome)
Babies with trisomy 18 have three copies of chromosome 18 and have severe intellectual disabilities and birth defects typically involving the heart, brain, and kidneys. Babies with trisomy 18 can also have visible birth defects such as an opening in the lip (cleft lip) with or without an opening in the roof of the mouth (cleft palate), a small head, clubbed feet, underdeveloped fingers, and toes, and a small jaw. Unfortunately, most pregnancies with trisomy 18 will miscarry. If born alive, most affected babies with trisomy 18 will pass away within the first few weeks of life. About 10 percent survive to their first birthday. Trisomy 18 occurs in approximately 1 in 3,000 live births.
Trisomy 13 (Patau syndrome)
Babies with trisomy 13 have three copies of chromosome 13 and have severe intellectual disabilities. They often have birth defects involving the heart, brain, and kidneys. Visible abnormalities include extra fingers and/or toes or an opening in the lip, with or without an opening in the palate. Given the severe disabilities, most pregnancies affected by trisomy 13 will miscarry. If born alive, most affected babies with trisomy 13 will pass away within the first few weeks of life. About 10 percent survive to their first birthday. Trisomy 13 occurs in approximately 1 in 5,000 live births.
SEX CHROMOSOME ANEUPLOIDIES**
Monosomy X (Turner syndrome)
Babies with monosomy X are biological females who have one X chromosome instead of two. Unfortunately, a high proportion of pregnancies with monosomy X will result in a miscarriage in the first or second trimester of pregnancy. Babies with monosomy X that make it to term may have heart defects, learning difficulties, and infertility. In most cases, babies with monosomy X will need extra medical care including hormone therapy at various stages of life.
XXY Syndrome (Klinefelter Syndrome)
Babies with XXY syndrome have two X chromosomes and one Y chromosome (XXY). This condition can be associated with learning difficulties and behavioral problems. People with Klinefelter syndrome might be infertile. About 1 in 500 biological males will be born with Klinefelter syndrome.
Triple X syndrome
Babies with Triple X syndrome have three X chromosomes (XXX). Children with this condition could be taller than average and might experience learning difficulties or behavioral problems. Approximately 1 in 800 biological females will be born with three X chromosomes.
XYY Syndrome (Jacob's Syndrome)
Babies with XYY syndrome have one X chromosome and two Y chromosomes (XYY). Most babies with XYY syndrome do not have any birth defects. Children with XYY could be taller than average and have an increased chance for learning, speech, and behavioral problems. Approximately 1 in 1,000 biological males will be born with one X chromosome and two Y chromosomes.
Complete Test Specifications for Singleton, Egg Donor, Gestational Carrier, and Monozygotic (Identical) Twins
Definition of performance terms
Sensitivity is the ability to correctly identify a truly positive case as positive. For example, in a group of Trisomy 21 cases, Vasistera™ NIPT will correctly identify more than 99% of those cases.
Specificity is the ability to correctly identify an unaffected case as negative.
Positive Predictive Value (PPV) is the likelihood the result says screen positive and the fetus is actually affected. For example, when Vasistera™ NIPT shows a screen positive result for Trisomy 21, there is a 95% chance that the fetus is affected by Trisomy 21. In other words, 5% of the time, you could get a screen positive result when the fetus is not affected by Trisomy 21.
Negative Predictive Value (NPV) is the likelihood the result says screen negative and the fetus is truly not affected.
| Condition | Sensitivity (95% CI) | Specificity (95% CI) | PPV | NPV |
|---|---|---|---|---|
| Trisomy 212,3 | 99.0% (CI 97.1-100) | >99% (CI 99.93-99.99) | 95% | >99.99%* |
| Trisomy 182,3 | 94.1% (CI 82.9-100) | >99% (CI 99.96-100) | 91% | >99.99%* |
| Trisomy 132,3 | >99% (CI 73.5-100) | >99% (CI 99.96-100) | 68% | >99.99%* |
| Monsomy X**3,4 | 94.7% (CI 74.0-99.9) | >99% (CI 99.7-100) | 78% | >99.99%* |
| XXX, XXY, XYY**5 | 73.1% (CI 61.0-85.1) | 99.9% (CI 99.90-99.99) | 83% | 99.87%* |
| Female | >99.9% (CI 99.4-100) | >99.9% (CI 99.5-100) | ||
| Male | >99.9% (CI 99.5-100) | >99.9% (CI 99.4-100) |
Test Specifications for Dizygotic (Nonidentical) Twins6
| Condition | PPV | NPV |
|---|---|---|
| Trisomy 21 | 88% | >99.975% |
| Trisomy 18 | 70% | >99.999% |
| Trisomy 13 | 67% | >99.999% |
References
1ACOG Practice Bulletin 226. Obstet Gynecol. 2020 Oct;136(4):859-867.
2Dar et al. Am J Obstet Gynecol. 2022 Aug;227(2):259.e1-259.e14.
3DiNonno et al. J Clin Med. 2019 Aug 26;8(9):1311.
4Martin et al. ISUOG World Congress 2022: September, 2022.
5Martin K et al. Genet in Med. 2023. https://doi.org/10.1016/j.gim.2023.100879
6Kantor et al. Prenat Diagn. 2022;42(13):1587-1593.
Footnotes
* Ongoing clinical follow-up is performed to ensure the NPV does not fall below the quoted value but follow up is not obtained for all low risk or screen negative calls.
** Not available for egg-donor or gestational carrier pregnancies or in cases of dizygotic (nonidentical) twins.