How does Vistara™ work?
Vistara™, single-gene NIPT, enables deeper insights for better health outcomes
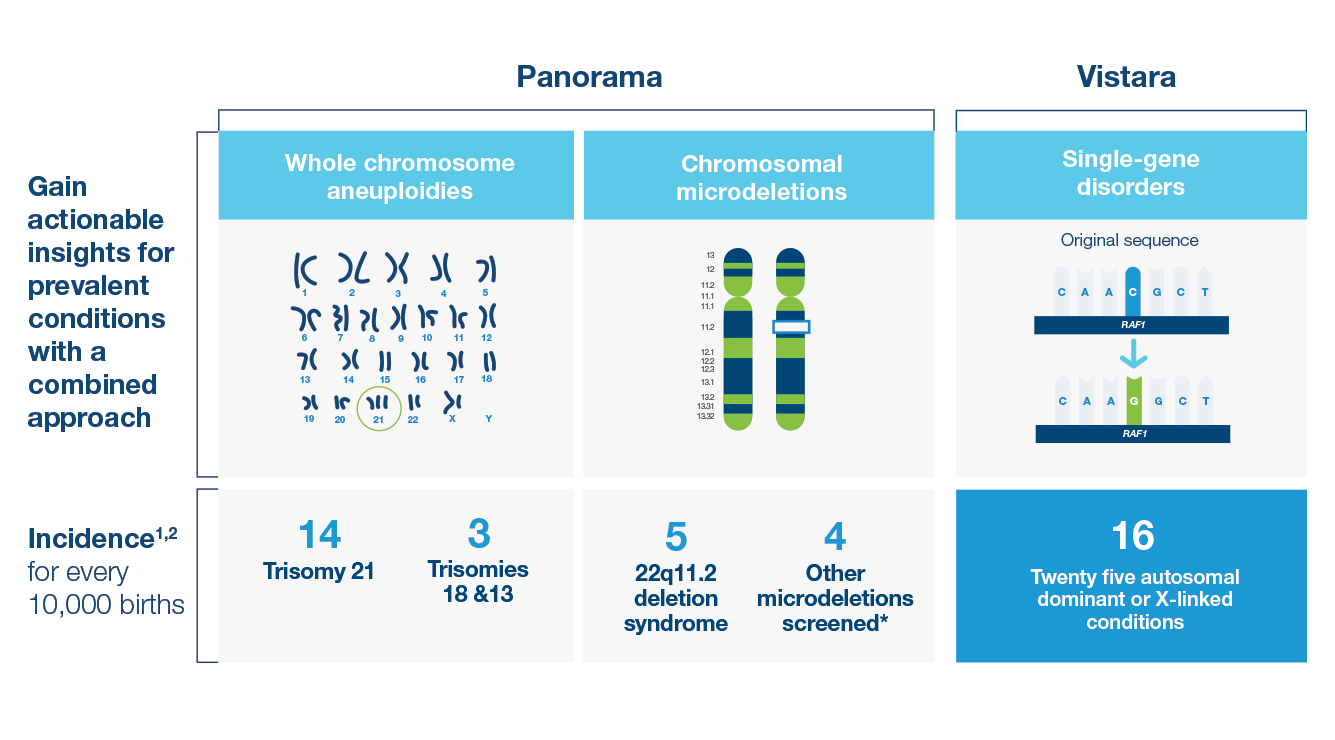
Early insights guide healthier futures
- Screen for 25 autosomal dominant and X-linked conditions across 30 genes with Vistara™ single-gene NIPT
- Combined incidence greater than Down syndrome1,2
- Provide clinically actionable information
- Many single-gene conditions benefit from early intervention
- Inform decisions for pregnancy and delivery management, early interventions, and patient education to ensure every child reaches their full potential
- Perform testing with ease and get clear results
- No paternal and/or egg-donor* sample necessary
- Find conditions other screening modalities would miss until after the window for critical intervention
*if applicable
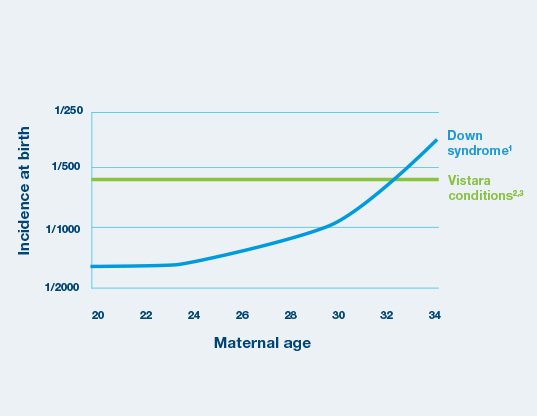
Vistara™ screens conditions with a combined incidence greater than Down syndrome
All pregnancies can benefit from Vistara™
Some scenarios where single-gene NIPT should be considered

Couples with a male partner over 40-years-old (advanced paternal age)
Ultrasound anomalies including shortened long bones and increased nuchal translucency

Family history of hereditary conditions

Patients who want as much information as possible
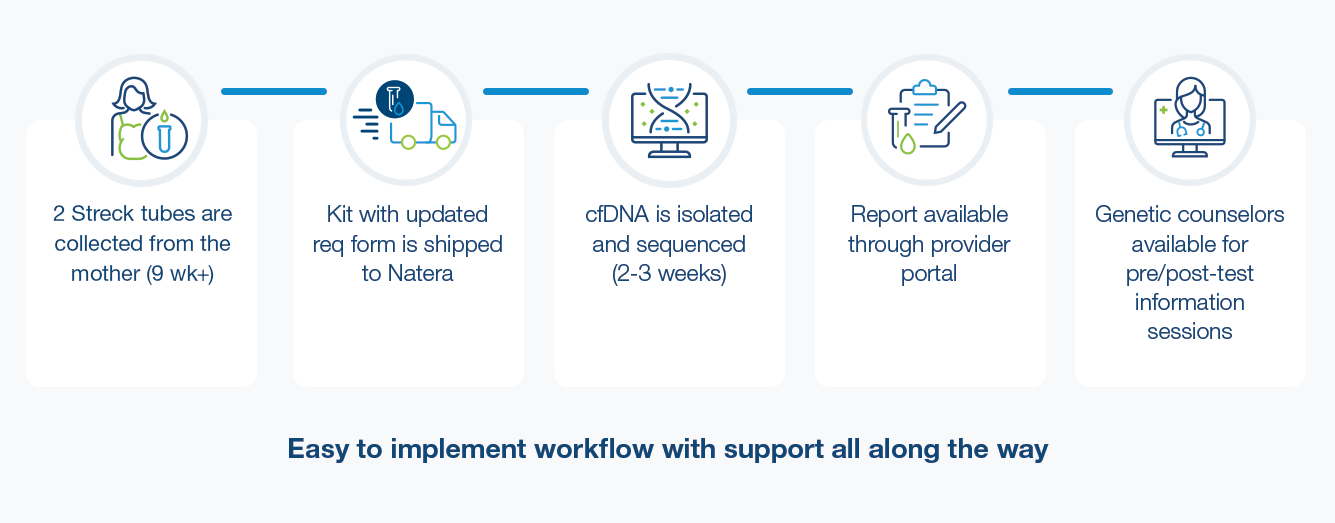
With only one blood-draw from the mother required, ordering Vistara™ is simple and can be integrated into any office scenario

No errors detected in clinical validation
Vistara™’s clinical validation published in Nature Medicine (Zhang et al) showed the following performance metrics:2
- >99% sensitivity
- >99% specificity
Make confident pregnancy management and early intervention decisions with performance you can trust
Vistara™ is a screening test, which means that Vistara™ does not make a final diagnosis. A screen-positive Vistara™ result means that a pregnancy could be affected by the condition that has screened positive. However, it cannot be known for sure if a baby has that condition based upon screening results alone. All medical decisions should be made after discussion with a heathcare provider regarding diagnostic testing during the pregnancy, like amniocentesis or chorionic villus sampling (CVS), or testing the baby after delivery.
Vistara™ is an incomparable adjunct to traditional diagnostic procedures like chorionic villus sampling (CVS) and amniocentesis with:
- One of the most comprehensive Noonan panels available
- Screening for conditions not included in standard diagnostic microarray
Findings on Vistara™ can be confirmed using the same original diagnostic sample, no need for additional confirmatory procedures**
**Cells must be saved for additional diagnostic testing
Is Vistara™ right for you?
We’re here to help you find out
References
1Snijders, et al. Ultrasound Obstet Gynecol 1999;13:167-170.
2Zhang J et al. Nat Med. 2019 Mar;25(3):439-447.
3Pagon RA et al., editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993-2017.