Inform Early Treatment Decisions
Identify High Risk Patients
to Guide Adjuvant Treatment
Test for presence of MRD after surgery to risk-stratify patients1
>27x
Higher risk of recurrence for ctDNA positive patients1
Detect
Recurrence Earlier
Detect recurrence
prior to standard imaging2
3 months
lead time in detecting recurrence compared to PET/CT2
Predict
Response to Treatment
Track changes in ctDNA levels to evaluate treatment response2,3
6 weeks
To identify patients with poor prognosis due to increasing ctDNA during 1L IO treatment3

Signatera™ treatment response monitoring is covered by Medicare for any patient being treated with immunotherapy

ctDNA testing is also recommended for assessment of disease burden in both virus-positive and virus-negative Merkel cell carcinoma (MCC)4

WEBINAR
Exploring tumor-informed ctDNA-guided interventions in melanoma
May 14th @ 5:00 PM ET | 2:00 PM PT
60 minute webinar
Upcoming Webinar
Transformative real-world insights and clinical applications from longitudinal tumor-informed ctDNA monitoring.
Join us for an insightful webinar exploring findings from a recent multi-center, real-world study, “Longitudinal ctDNA Monitoring for Post-Surgical Disease Surveillance in Patients with Stage I–IIIB Melanoma.”
Key topics include:
- Tumor-informed ctDNA as a biomarker: Learn how ctDNA detection can provide early insight into melanoma recurrence and progression
- Real-world evidence: Explore study findings on tumor-informed ctDNA-guided interventions and their potential to improve patient outcomes
- Clinical applications: Understand how Signatera™ results may guide care decisions, including imaging escalation, therapy initiation, and management of molecular recurrence
Inform Your Skin Cancer Treatment Approach With Signatera™
Risk-stratify early-stage patients to inform adjuvant treatment decisions
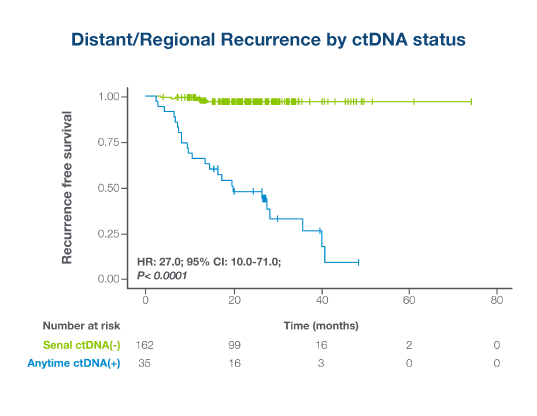
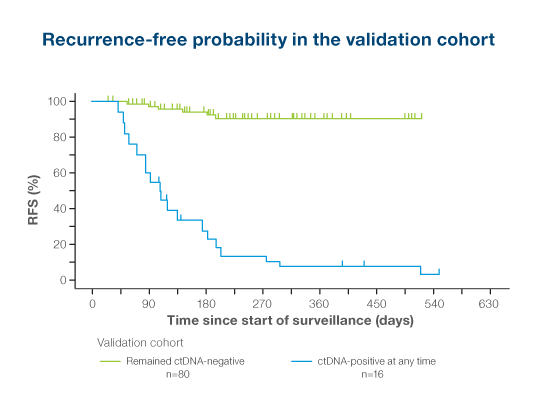
ctDNA-positivity was the most significant prognostic factor associated with RFS in multivariate analysis among stage I-IIIb resected melanoma patients1
- ctDNA-positivity at any postoperative timepoint was significantly associated with shorter RFS (HR: 27.0; P < 0.0001)
- ctDNA-positivity during surveillance monitoring identified patients with 24x higher risk of recurrence
- Signatera™-positivity influenced changes in treatment management in 73.7% of patients
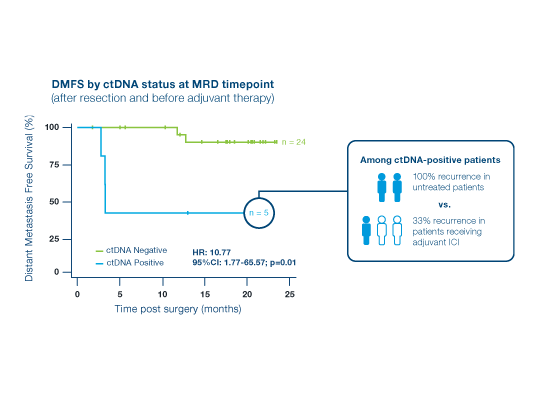
Inform adjuvant treatment decisions and detect recurrence early
Patients with detectable ctDNA at one timepoint after surgery had a 10x higher risk of recurrence than ctDNA-negative patients2
- ctDNA-positivity was associated with significantly shorter DMFS (HR=10.77; p=0.01)
- ctDNA-positivity at week 6 of adjuvant ICI therapy was significantly associated with an inferior DMFS (HR: 34.54, P<0.0001)
- In longitudinal analysis, Signatera™ provided a lead time to recurrence of 3 months vs. PET/CT imaging (average time to recurrence of ~6 months vs ~9 months)
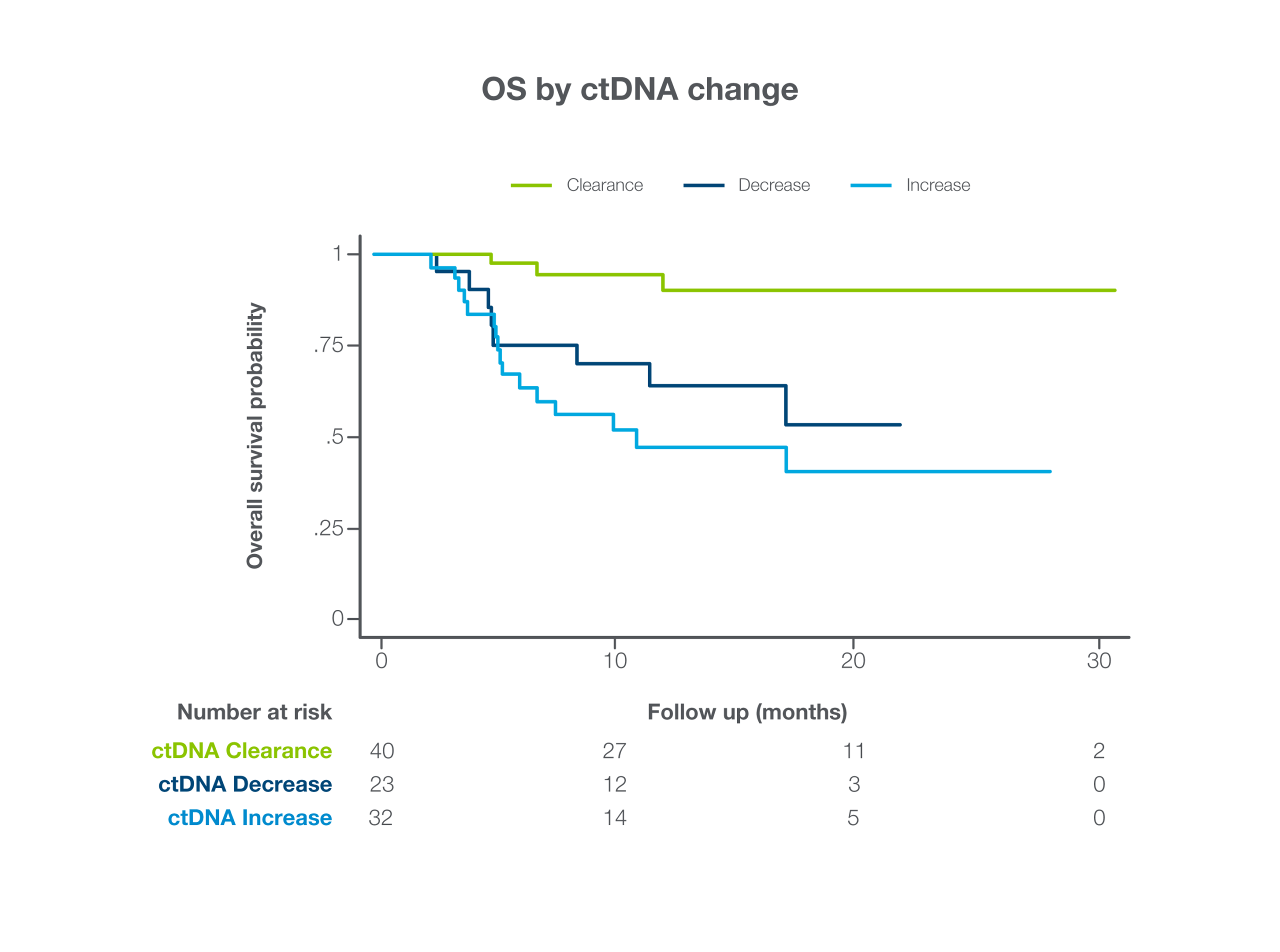
Predict response to ICI treatment
Signatera™ ctDNA dynamics 6-8 weeks after ICI treatment initiation may inform earlier treatment decisions3
- Patients with ctDNA clearance had improved PFS (HR = 5.15, p < 0.001) and OS (HR = 5.72, p = 0.010) compared to patients with decreasing but still positive ctDNA
- Patients with ctDNA increase had shorter PFS (HR = 5.67, p < 0.001) and OS (HR = 8.76, p = 0.001)

Inform Merkel Cell Carcinoma surveillance strategy
Circulating tumor DNA (ctDNA) is now recommended by the National Comprehensive Cancer Network (NCCN®) for assessment of disease burden in both virus-positive and virus-negative Merkel Cell Carcinoma (MCC)4
- Merkel Cell Carcinoma represents the first instance of an NCCN recommendation for ctDNA MRD testing in a solid tumor cancer, a significant step forward in improving patient management4
- ctDNA was recently added as an NCCN Category 2A recommendation to the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for both virus-positive and virus-negative patients across all stages4
- Testing is recommended as part of an additional workup during the initial diagnosis and as part of routine follow-up surveillance4
- The NCCN Guidelines® indicated that testing is “often obtained every 3 months.”4
Recurrence Detection in Merkel Cell Carcinoma
Patients who were Signatera™-positive had up to a 20x higher risk of recurrence (HR:20)5
- Signatera™ demonstrated 95% sensitivity for detecting disease at enrollment
- ctDNA-positive patients had >70% likelihood of recurrence within 6 months of a positive test, while ctDNA-negative patients had a <5% likelihood of recurrence within 3 months of a negative test
- Signatera™ demonstrated a 94% PPV and 93% NPV for recurrence respectively
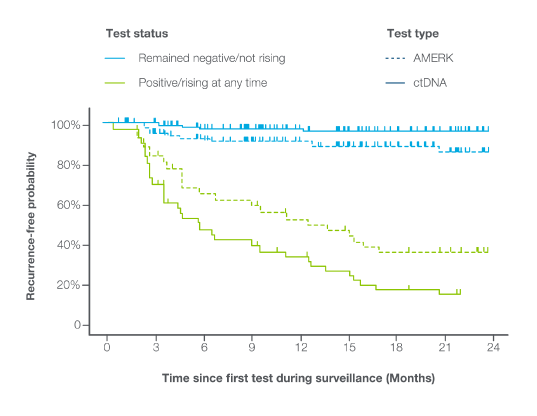
Signatera™ vs AMERK
Signatera™ ctDNA demonstrated superior prognostic value for recurrence compared to AMERK (HR = 37.2 vs HR = 6.5)6
- Signatera™ detected recurrence more frequently and earlier than AMERK, often preceding imaging
- Signatera™ detected >80% of the recurrences missed by AMERK
- Signatera™ demonstrated higher positive and negative predictive value than AMERK at all timepoints
Provide Confidence for Patients With Skin Cancer
After Jeffrey started dual immunotherapy for his metastatic melanoma, Signatera™ showed a dramatic drop in his ctDNA levels and helped provide assurance that his treatment was working.
Watch Jeffrey’s story to learn how his oncology team used Signatera™ to help guide his care and restore his confidence in his future.
MRD Testing for Melanoma: The Next Step in Recurrence Prevention
Watch this webinar from the Melanoma Research Foundation to learn more about how MRD is being used to help manage melanoma.
*Clicking this button will open it on YouTube.com
Learn More About Signatera™ in Skin Cancers
Is Signatera™ for skin cancer right for your patients?
1Ansstas G, et al. Presented at the ASCO Annual Meeting, Chicago, IL, 2025.
2Eroglu Z, et al. Cancer, 2023. https://doi.org/10.1002/cncr.34716
3Burkey, et al. Presented at the ASCO Annual Meeting, Chicago, IL, 2025.
4Referenced with permission from the NCCN Clinical Practice Guidelines In Oncology (NCCN Guidelines®) for Merkel Cell Carcinoma V.1.2026 © National Comprehensive Cancer Network, Inc. 2025. All rights reserved. Accessed September 2, 2025. To view the most recent and complete version of the guideline, go online to NCCN.org. NCCN makes no warranties of any kind whatsoever regarding their content, use or application and disclaims any responsibility for their application or use in any way.
5Akaike T, et al. Journal of Clinical Oncology, 2024.
6Chan J, et al. Poster presented at ASCO Annual Meeting, Chicago, IL, 2025.




