New Medicare Coverage: Signatera in NSCLC
Learn MoreNew Medicare Coverage: Signatera in NSCLC
Learn MoreDetect and Monitor ctDNA in NSCLC to Inform Treatment Strategy
Identify
High Risk Patients
10x higher
risk of recurrence for patients testing ctDNA-positive with Signatera™ Genome between surgery and adjuvant therapy1
Detect
Recurrence Earlier
>5 months
median lead time to recurrence vs standard imaging2,3
Monitor Immunotherapy
Treatment Response
96% lower
Risk of death for patients who cleared their ctDNA by week 9 compared to patients with increasing ctDNA4
Better Biomarkers are Needed to Optimize NSCLC Patient Care
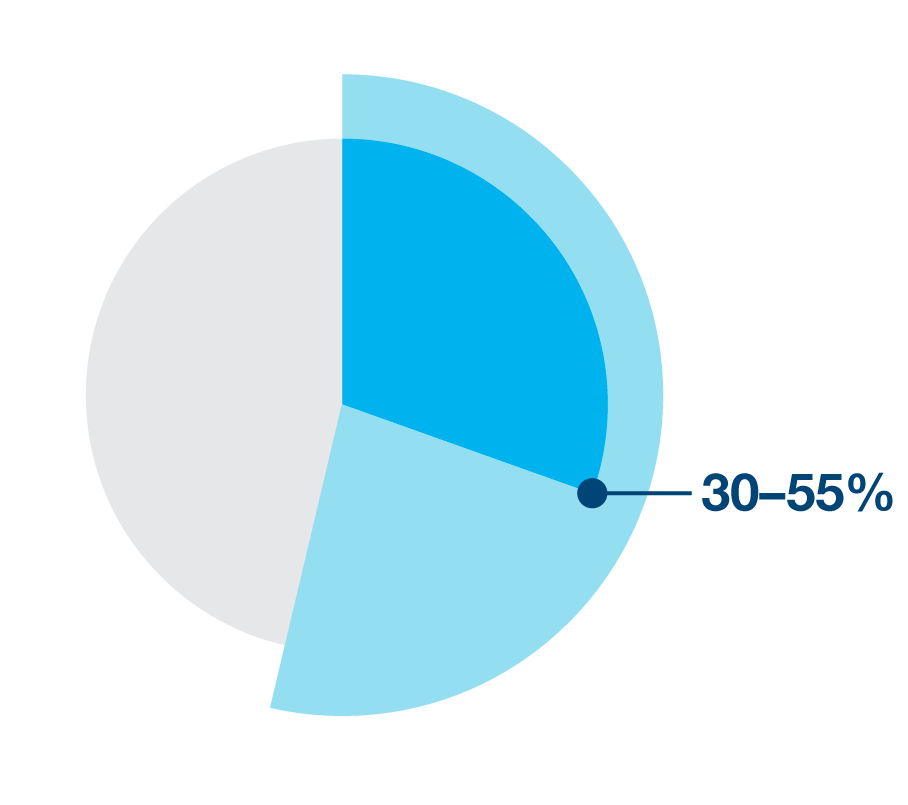
30-55% of NSCLC patients will develop disease recurrence post-definitive treatment (surgery; chemo-radiation)5
Which stage I-III patients require adjuvant or extended therapy may be unclear
Early detection of recurrence with less tumor burden may enable local treatment with curative-intent, potentially improving outcomes6
Inform Your Lung Cancer Treatment Approach With Signatera™
Monitoring circulating tumor DNA (ctDNA) with Signatera™ across the treatment journey can answer important clinical questions and inform treatment decisions.
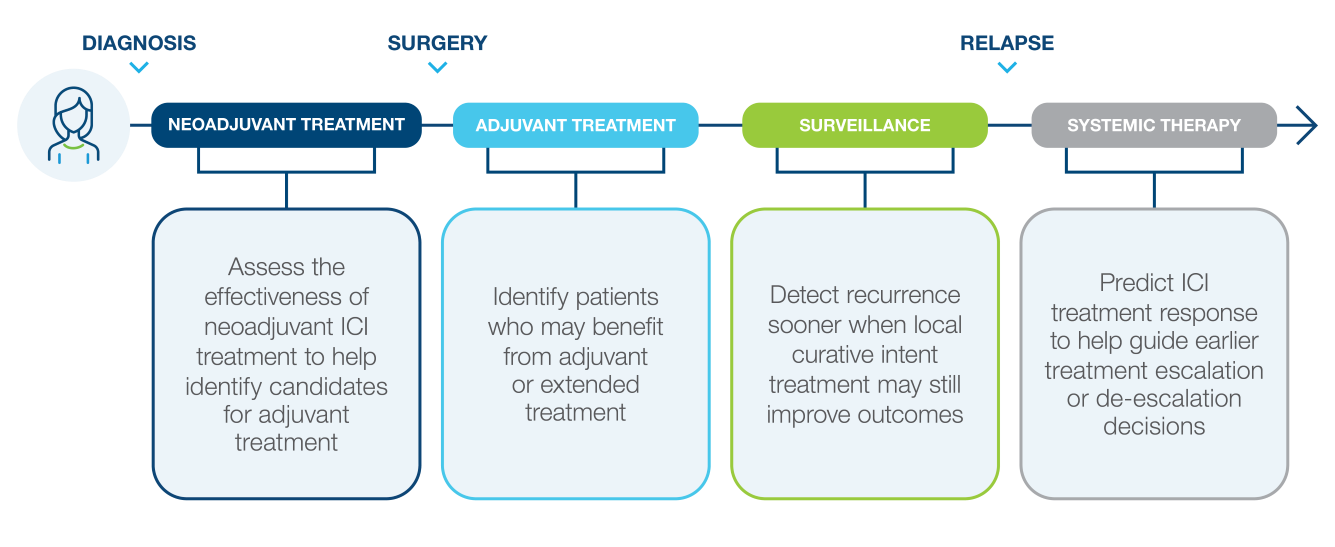
Identify high risk patients after curative intent treatment
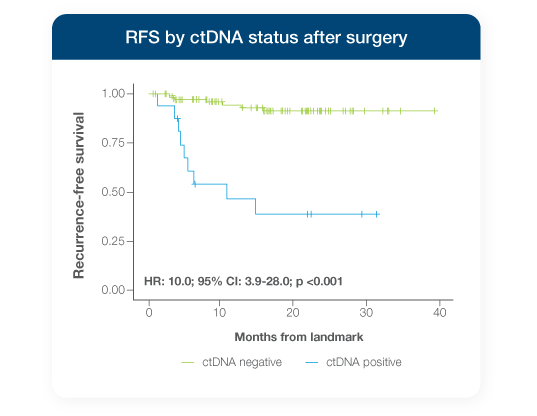
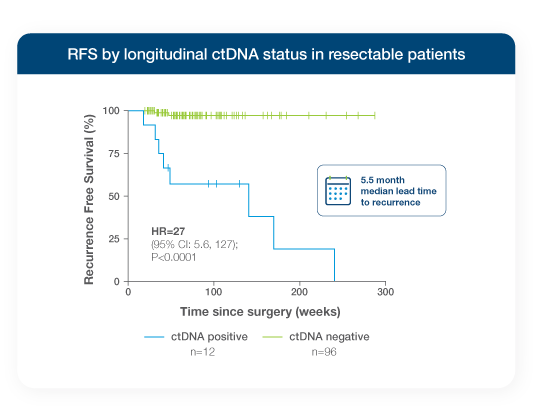
Stage I-III resectable NSCLC patients testing ctDNA-positive with Signatera™ Genome between surgery and adjuvant therapy had a 10x higher risk of recurrence1
- Signatera™ Genome ctDNA-negativity during the MRD window (post-surgery, pre-adjuvant therapy) was associated with improved RFS & OS1
- Every patient who tested ctDNA-negative after surgery remained alive through 18.8-month median follow-up, whereas ctDNA-positive patients achieved just 57.5% 24-month OS1,7
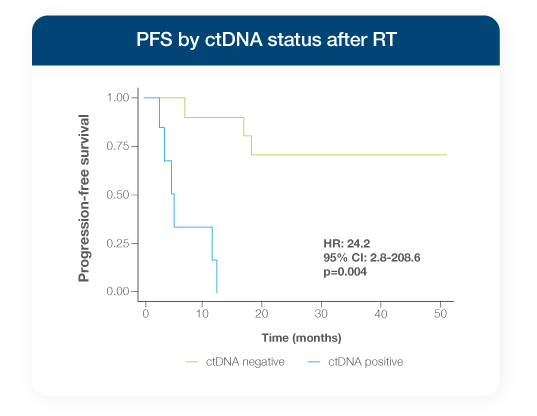
Stage I-III unresectable NSCLC patients testing ctDNA-positive with Signatera™ at a single timepoint after radiation therapy (RT) had a >24x higher risk of progression2
- ctDNA-positivity at the first post-RT time point was highly prognostic of progression2
- Every patient who tested ctDNA-positive went on to recur2
Detect recurrence early
Signatera™ detected recurrence a median of >5 months earlier than standard imaging across resectable and unresectable disease2,3
- Every patient experiencing extracranial recurrence tested ctDNA-positive with Signatera™3
- 98% of patients testing ctDNA-negative did not recur through follow-up3
- Post-operative radiographic surveillance strategies were altered in 100% of ctDNA-positive patients (earlier radiographic imaging), and patients with PET scans positive for malignant features received early referrals for treatment3
Evaluate Response to ICI Treatment in NSCLC
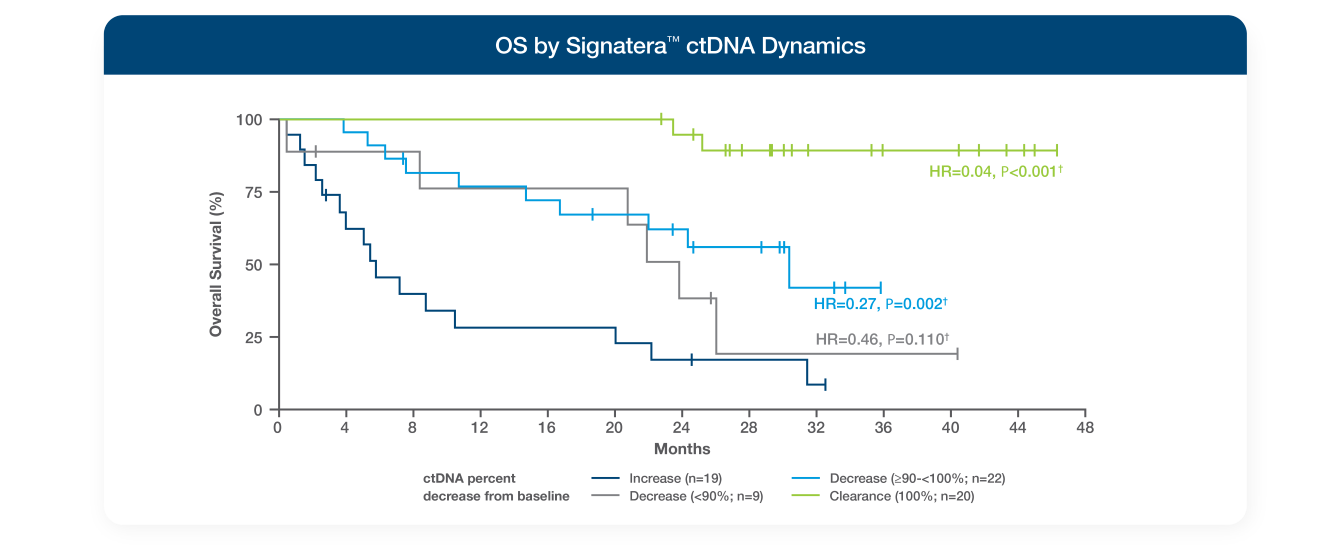
Signatera™ was evaluated in the EMPower Lung-1 trial, a prospective study in patients with advanced or metastatic NSCLC randomized to immune checkpoint inhibitor (ICI) vs chemotherapy for 1L treatment4
- Rising ctDNA by week 3 or lack of ctDNA clearance by week 9 identified patients experiencing inferior clinical outcomes4
- ctDNA deep decrease (>90%) and clearance were associated with significantly improved OS4
- Composite ctDNA & RECIST assessment may improve prediction of OS benefit from ICI4
Watch to Learn More About Signatera™
See the Latest Data and How Signatera is Used to Personalize NSCLC Patient Care with Oleg Gligich, MD and Michael Krainock, MD, PhD
Review the EMPOWER-Lung 1 trial with Natalie Vokes, MD
Reassure Your Patients Living With Lung Cancer
After JoAnn was diagnosed with lung cancer, Signatera™ detected a rise in her ctDNA levels, alerting her oncologist to a progression that was not scannable.
Watch JoAnn’s story to learn how she and her oncologist added Signatera™ testing to their toolbelt to help closely monitor her cancer.
“I want to know. The more you know, the better off you are.” – JoAnn, living with lung cancer

Do More With Less Tissue
One tumor sample – two tests
Lung Cancer Resources

The benefits of using ctDNA over traditional scans to assess responses to ICI
Dr. Natalie Vokes shares thoughts on how ctDNA can be used in addition to scans in NSCLC

Signatera™ in Lung Cancer
Learn about Signatera™ for risk-stratification, surveillance, and immunotherapy response monitoring

Patient Case Study
Read how Signatera™ detected a rise in ctDNA levels during immunotherapy, informing the decision to pivot to a combination therapy approach.
Is Signatera™ for lung cancer right for your patients?
*OS = Overall Survival
References
1Becherano G. Presented at the IASLC 2025 World Conference on Lung Cancer. September 2025, Barcelona, Spain.
2Lebow E, et al. Frontiers in Oncology. 2023;13:1253629.
3Martin T, et al. Journal of Thoracic and Cardiovascular Surgery. 2024.
4Vokes N, et al. Poster presented at the ASCO Annual Meeting, Chicago, IL. June 2023.
5Uramoto H, Tanaka F. Translational Lung Cancer Research. 2014;3:242–249.
6Gomez DR, et al. Journal of Clinical Oncology. 2019;37:1558–1565.
7Natera Data on File.