Evaluate treatment response sooner
Predict response
to immunotherapy after just 2 cycles
98%
of patients with an increase in ctDNA at the beginning of cycle 3 did not derive an objective response to immunotherapy treatment.¹
Identify
exceptional responders
100%
OS in patients who cleared ctDNA for at least one on-treatment time point (median follow-up beyond first clearance of 25.4 months)¹
Longitudinal monitoring with Signatera™ helps answer critical questions
Signatera™ quantifies ctDNA over time to provide a real-time assessment of changes in disease burden during immunotherapy (IO) treatment.¹

Discover the treatment response monitoring data

INSPIRE trial: Predict immunotherapy response across solid tumors
The INSPIRE trial validated Signatera™ ctDNA testing as an early biomarker of immunotherapy response with 98% baseline ctDNA detection across 94 patients and 25 solid tumor types.¹

Melanoma: Inform risk-based treatment selection and monitor immunotherapy response
69 advanced melanoma patients monitored across 555 plasma timepoints to determine if ctDNA can help risk-stratify patients for disease recurrence and predict response to immunotherapy.²
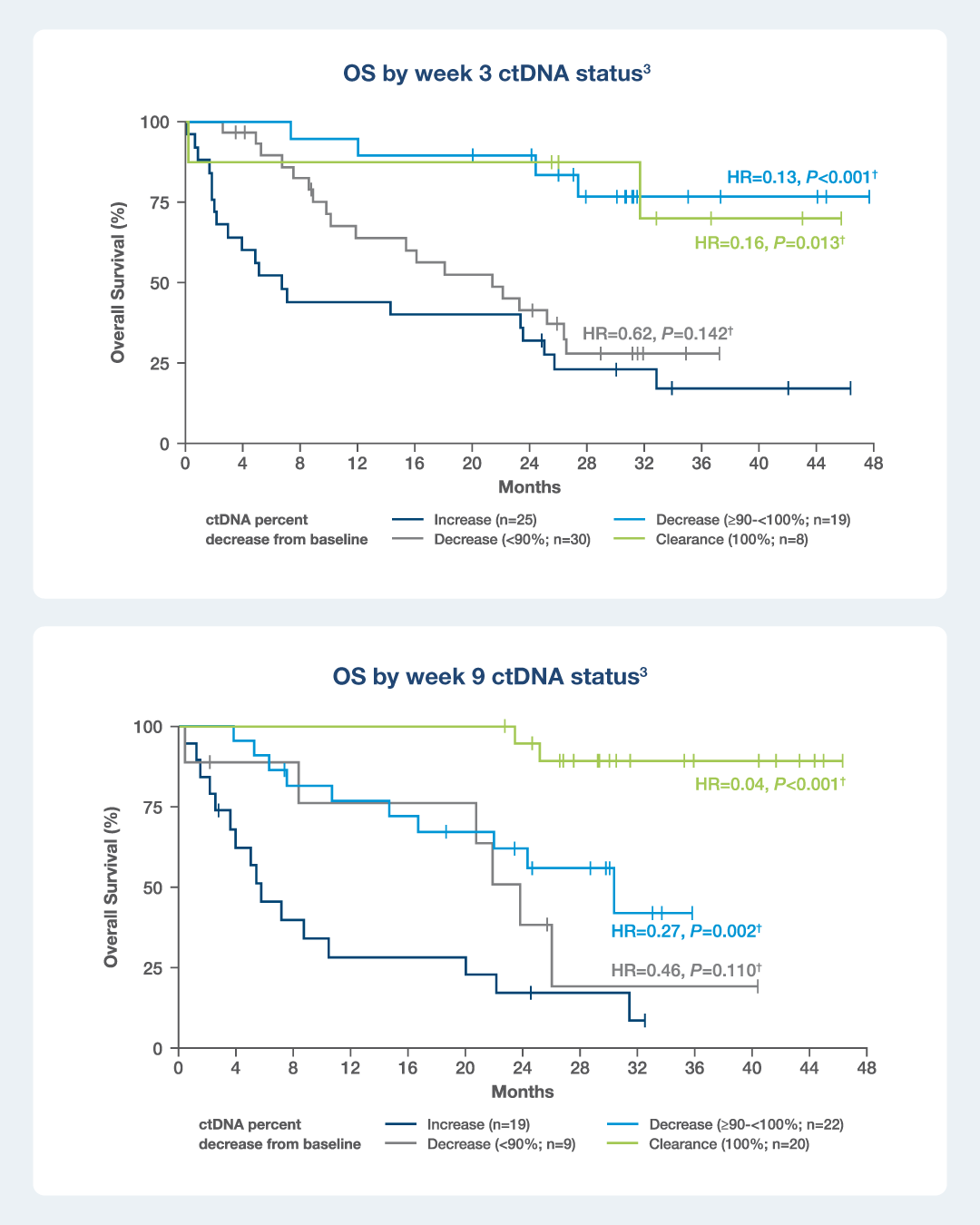
NSCLC: Inform early treatment escalation on immunotherapy
Signatera™ was evaluated in the EMPower Lung-1 trial, a prospective study in patients with advanced or metastatic NSCLC randomized to ICI vs chemotherapy for 1L treatment.³
Learn how Signatera™ MRD testing informed the treatment strategy for a patient fighting melanoma.
“Signatera™ gives us confidence with how we think about what’s going on with the patient … their prognosis and whether or not we are doing the right thing for them.”
– Steven Liu, MD, medical oncologist

Comprehensive patient insights and Molecular Residual Disease Detection from the same tumor tissue
Altera™ empowers personalized therapy selection by identifying treatments most likely to benefit patients based on their tumor biomarkers and cancer type. The comprehensive report covers both well-established cancer driver genes and less common but actionable biomarkers, giving clinicians deeper insights to guide informed treatment decisions. Combine with Signatera™ MRD testing using the same tissue to optimize patient management throughout the treatment journey.
Is Signatera™ right for your patients on immunotherapy?
1Bratman SV, Yang SYC, Iafolla MAJ, et al. Personalized circulating tumor DNA analysis as a predictive biomarker in solid tumor patients treated with pembrolizumab. Nature Cancer. 2020;1(9):873-881.
2Eroglu Z, Krinshpun S, Kalashnikova E, et al. Circulating tumor DNA based molecular residual disease detection for treatment monitoring in advanced melanoma patients. Cancer. 2023. https://doi.org/10.1002/cncr.34716.
3Vokes N, et al. Circulating Tumor DNA (ctDNA) Dynamics and Survival Outcomes in Patients with Advanced NSCLC and High (>50%) PD-L1 Expression, Randomized to Cemiplimab vs Chemotherapy. Presented at ASCO Annual Meeting, Chicago, IL, June 2023.
4Powles T, Assaf ZJ, Davarpanah N, et al. ctDNA guiding adjuvant immunotherapy in urothelial carcinoma. Nature. 2021.
5Powles T, Assaf ZA, Degaonkar V, et al. Updated Overall Survival by Circulating Tumor DNA Status from the Phase 3 IMvigor010 Trial: Adjuvant Atezolizumab Versus Observation in Muscle-invasive Urothelial Carcinoma. European Urology. 2023; https://doi.org/10.1016/j.eururo.2023.06.007.