Personalize your lung cancer care

Understand your risk
Signatera™ can predict your risk of lung cancer recurrence after surgery or radiation.1,2

Detect recurrence early
Signatera™ has been shown to detect lung cancer recurrence >5 months before standard scans.1,2

Know if immunotherapy is working
Signatera™ can predict if your lung cancer is responding to immunotherapy as early as 3 weeks into treatment.3
Hear from a Signatera™ lung cancer patient
After JoAnn was diagnosed with lung cancer, she learned how Signatera™ could help her doctor detect potential disease worsening ahead of scans.
“Signatera™ is a great warning system that something is going on. From there, you figure out what it is. Signatera™ does empower me to be my own best advocate with my oncologists.” – JoAnn, living with lung cancer

Be your own best advocate
Ask your doctor about Signatera™
If you or your loved one have been diagnosed with lung cancer, you likely have many questions including “What do I do next?” When used alongside other tests, Signatera™ can help answer critical questions to guide your care:
- Is there cancer left after my surgery or radiation?1,3
- Is my cancer coming back?2,3
- Is my immunotherapy working?3
Access testing
Billing
- Signatera™ is covered by Medicare for stage I-III non-small cell lung cancer (NSCLC) in the surveillance setting, as well as for any patient being treated with immunotherapy.
- We welcome all insurance plans. Please refer to our list of in-network plans that we participate with, or call your insurance provider.
- We offer financial assistance programs for those that qualify.
How does Signatera™ testing work?
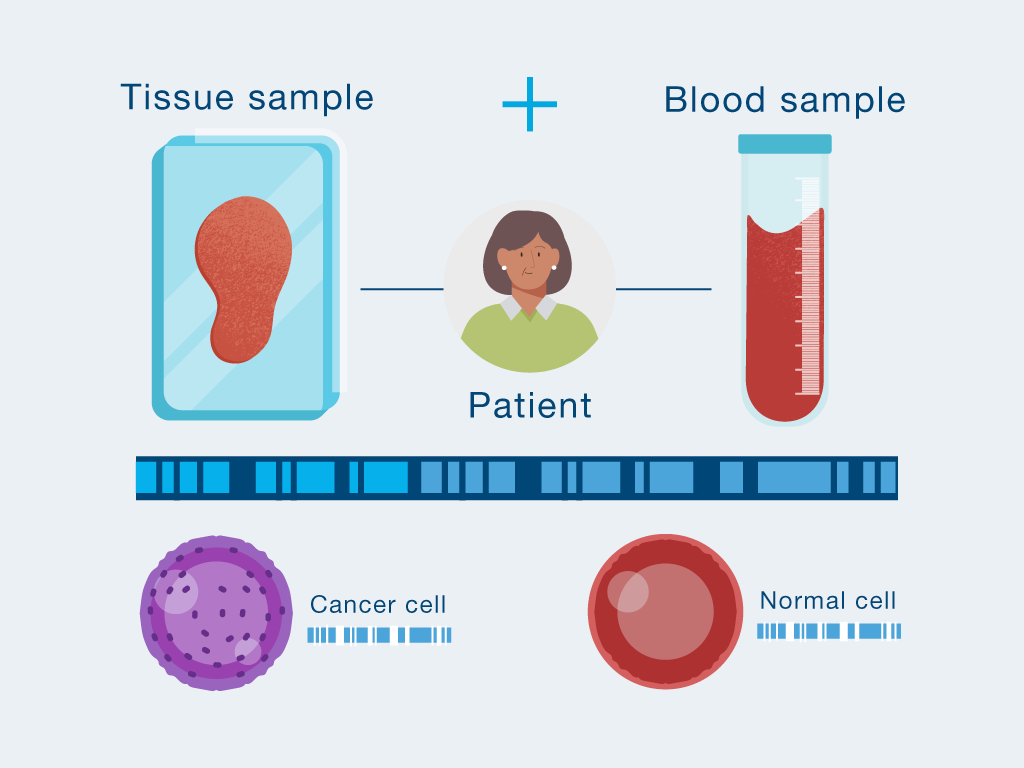
Personalized, tumor-informed test created
The first time your doctor orders Signatera™, a one-time tissue sample and a blood sample are needed to build your unique test. Natera will work with your cancer care team to get your tumor tissue from a procedure or surgery you had in the past.

Blood draw checks for tumor DNA
After your test is built, you only need to provide a blood sample each time your doctor orders Signatera™.
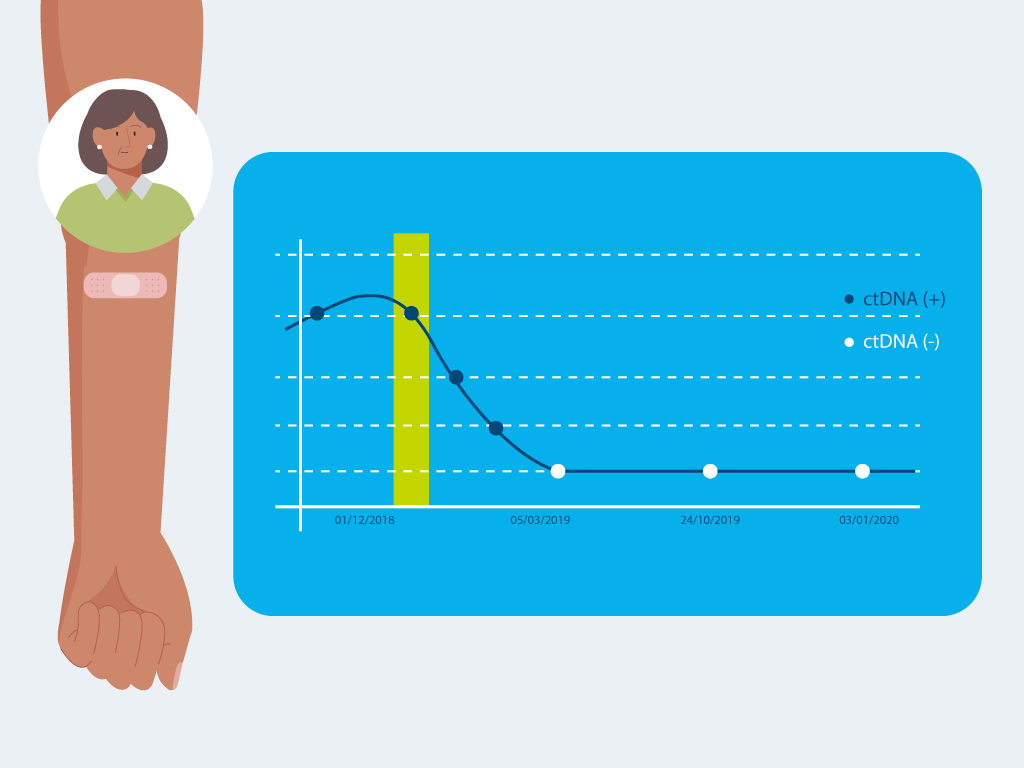
Designed for monitoring over time
Repeated Signatera™ testing can show changes in your ctDNA levels, helping your doctor understand if your cancer is shrinking, growing, or coming back.
Genetic testing for lung cancer patients
In addition to personalized cancer monitoring with Signatera™, genetic testing plays a vital role in lung cancer care. The Altera™ Tumor Profiling Test can help determine if targeted or immunotherapy will be effective by analyzing genes like EGFR and ALK. This personalized approach is especially important for non-small cell lung cancer (NSCLC) patients.
By combining Signatera™ with Altera™, lung cancer patients can personalize both their current therapies and future care, ensuring their treatment is tailored to their genetic profile.
Common questions about lung cancer and Signatera™
What is ctDNA and what does it mean in lung cancer?
ctDNA stands for circulating tumor DNA. These are small pieces of DNA that can come from cancer cells and may be found in the blood. A ctDNA blood test looks for that tumor DNA signal to add information during monitoring and follow-up.
What is Signatera™?
Signatera™ is Natera’s personalized ctDNA blood test, also called an MRD test. It is built using your tumor tissue, so it is designed to look for DNA that matches your cancer’s unique fingerprint over time.
Does Signatera™ replace CT scans or PET scans?
No. Imaging and clinic visits remain essential. Signatera™ is designed to add another source of information, not replace scans, exams, or other lab tests.
Why does this page focus more on non-small cell lung cancer (NSCLC)?
Most of the published Signatera™ lung cancer evidence and the Medicare surveillance coverage information Natera shares are focused on NSCLC. That is why NSCLC is highlighted more in the research and coverage sections.
If I have small cell lung cancer (SCLC), can ctDNA monitoring still be discussed?
Possibly. Ask your care team whether tumor-informed ctDNA monitoring fits your treatment plan. Natera describes Signatera™ for immunotherapy response monitoring across solid tumors, which may be relevant for some SCLC patients receiving immunotherapy. Your doctor will decide how to use ctDNA results alongside imaging and symptoms.
What does a positive Signatera™ result mean in lung cancer follow-up?
A positive result means ctDNA was detected at that timepoint. In NSCLC studies, ctDNA detection after treatment has been associated with higher recurrence risk and outcomes over time. A positive result does not confirm a visible recurrence by itself. Your doctor will interpret it with imaging, exams, and your full history.
What does a negative Signatera™ result mean?
A negative result means ctDNA was not detected at that timepoint. This can be reassuring, but it does not guarantee the cancer will not return. Your doctor will still recommend routine follow-up.
How often is Signatera™ ordered in lung cancer?
There is not one schedule for everyone. Some doctors use serial monitoring, meaning repeat testing over time, based on stage, treatment plan, and follow-up schedule.
Why might my doctor also order tumor profiling?
Tumor profiling can look for genetic changes and biomarkers that may help guide targeted therapy, immunotherapy decisions, or clinical trial discussions. In lung cancer, this is most often discussed in NSCLC. Natera offers tumor profiling through Altera™.
Is Signatera™ right for you?
1Martin T, Dinerman A, Sudhaman S, et al. Early real-world experience monitoring circulating tumor DNA in resected early-stage non-small cell lung cancer. Journal of Thoracic and Cardiovascular Surgery. 2024.
2Lebow E, et al. Frontiers in Oncology. 2023;13:1253629.
3Vokes N, et al. Poster presented at ASCO Annual Meeting, Chicago, IL, June 2023.