How Can You Evaluate NIPT Performance?
Last year, the American College of Obstetricians and Gynecologists (ACOG) updated its prenatal screening guidelines to recommend that all pregnant women should have access to noninvasive prenatal testing (NIPT), citing evidence that NIPT is the most accurate screening method for common aneuploidies.1
Even in average risk pregnancies, NIPT utilization and the development of new NIPT technologies will likely continue to grow. As NIPT adoption increases, it is important that we have standards for evaluating NIPT performance across technologies to ensure that we are offering patients the best possible care through the highest-quality, most-reliable screens.
One key reporting variable for NIPT performance is positive predictive value (PPV). As PPV is applied as a tool to evaluate NIPT, it is becoming clear that PPV is a reliable metric of NIPT quality, and that it offers a means to evaluate NIPT performance across patient populations.2
What is PPV, and why is it important?
In the context of NIPT, PPV tells us how likely it is that someone with a positive NIPT test result truly has an affected pregnancy. In other words, PPV gives us an idea of the extent to which we can expect test results to match reality. For example, the Panorama NIPT has a PPV of 95% for trisomy 21, which means that if a patient tested with Panorama has a high-risk result for trisomy 21, there is approximately a 19 out of 20 chance that the baby has the chromosomal abnormality.3
PPV can be calculated as true positives divided by the sum of true positives and false positives: true positives/(true positives + false positives)
A PPV calculation can be performed based on real-world data or analytical data, and the data type used affects the value and meaning of the calculated PPV. Panorama relies on clinical PPVs, rather than analytically determined PPV calculators or “personalized” PPVs, for several reasons, including that only clinical PPVs are derived from patient outcomes.
Clinical PPVs are based on clinical outcomes studies that occur in clinical practice. On the other hand, analytical PPV calculators extrapolate data from validation studies that could exclude important factors such as vanished twins or maternal chromosome abnormalities. Because these factors can alter a test’s specificity, if such factors are excluded, the PPVs could be overestimated.
Another advantage of clinical PPVs is that they can reflect the performance of complex algorithms for which simple analytical calculators cannot account. For example, the Panorama algorithm incorporates both maternal and gestational age into its aneuploidy risk assessment. Analytical PPV calculators cannot account for these factors, because they assume NIPT results are independent of prior risk. Therefore, it is inappropriate to use a Panorama result in a PPV calculator, because doing so would account for maternal age risk twice.
What do we know about NIPT PPVs?
The largest NIPT outcomes study to date provides some insight into the value of PPVs for assessing NIPT performance. The study cohort included more than 1 million patients, of which 13,231 (1.3%) were considered high-risk for common aneuploidies. The study analyzed the clinical performance of Panorama as part of a rigorous quality assurance program.2 Importantly, the results showed that PPV did not differ significantly for older versus younger populations, which is in contrast to the analytical PPVs of counting based tests.
Figure 1: Panorama trisomy 21 clinical PPVs are stable across age groups while “counting” NIPT analytical PPVs are much lower in younger women2–5
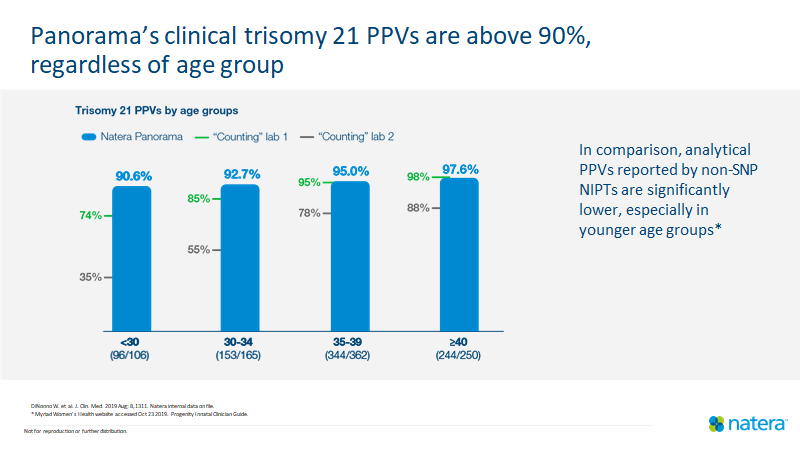
Figure 2: Panorama’s clinical PPVs for the common aneuploidies were comparable between average-risk and high-risk patients2
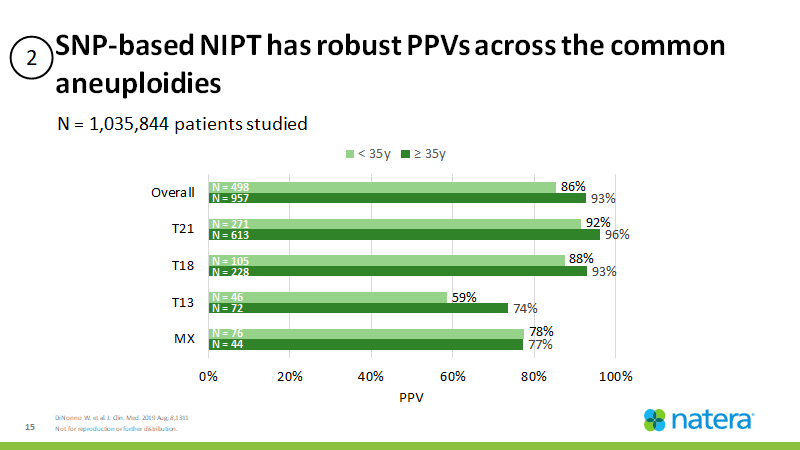
These high PPVs support broadening the use of SNP-based NIPT to younger women of average risk
Why is Panorama PPV stable at lower age groups?
Given the past concern that NIPT results would be significantly more reliable in older, higher risk women, resulting in a lower PPV in younger women, it is important to reflect on the observed stability of PPV in younger populations. Multiple potential explanations are possible.
For instance, Panorama’s SNP-based NIPT is associated with lower false positive rates than other NIPTs because Panorama can detect biological phenomena that are common causes of false positives in counting-based tests. These phenomena include vanishing twins and maternal chromosomal abnormalities.
Additionally, Panorama’s algorithm incorporates maternal age-based prior risk into its risk score. Younger women also could have lower false positive rates because of their lower risk of aneuploidy, and therefore lower risk of confined placental mosaicism, a known-source of NIPT false positives.1
PPV offers a way to evaluate NIPT technologies and establish performance standards
SNP-based NIPT has demonstrated consistent test performance in terms of sensitivity, specificity, and PPV. The newest data that show the stability of PPV across populations suggest that NIPT should be offered to all pregnant women, and that PPV can serve as a test quality assurance measure. We should therefore use clinical PPV to evaluate the performance of specific NIPT technologies and the value of NIPT for subpopulations of pregnant women.
Learn more about the only SNP-based NIPT, Panorama, here: https://www.natera.com/womens-health/panorama-nipt-prenatal-screening/
Natera is committed to helping women access Panorama and believes that the cost of your prenatal screening test bill should not be a barrier. If you have questions about what you might expect on your prenatal screening test bill, please contact the Natera billing phone number at 1-844-384-2996 (8 am-7 pm CT M-F) or visit the Natera billing page.
References
- Screening for Fetal Chromosomal Abnormalities. Obstetrics & Gynecology. 2020;136(4):859-867. doi:10.1097/AOG.0000000000004107
- DiNonno W, Demko Z, Martin K, et al. Quality Assurance of Non-Invasive Prenatal Screening (NIPS) for Fetal Aneuploidy Using Positive Predictive Values as Outcome Measures. Journal of Clinical Medicine. 2019;8(9):1311. doi:10.3390/jcm8091311
- Myriad Women’s Health website accessed Oct 23 2019.
- Progenity Innatal Clinician Guide.
- Natera internal data on file.
