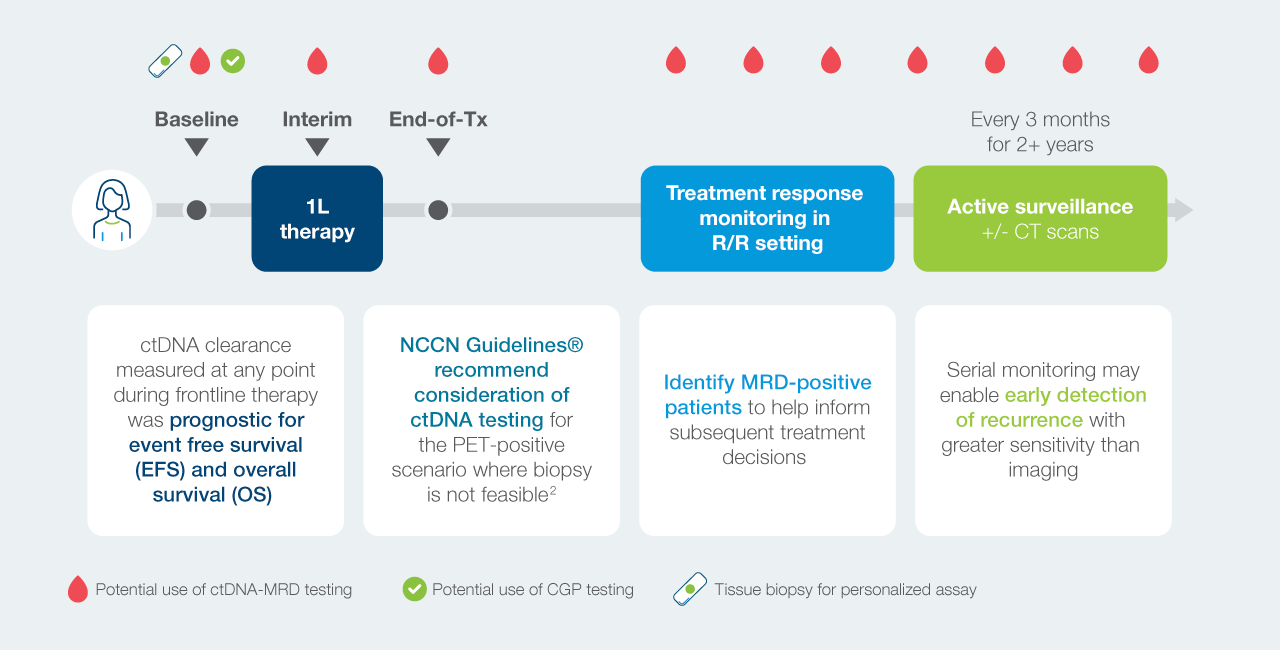
Current limitation in lymphoma care where ctDNA can play an instrumental role in transforming patient management
High relapse risk
Lymphoma is the 6th most common cancer in the U.S. and has high relapse rates after frontline chemotherapy.
Monitoring limitations
PET/CT is the standard for response assessment but has high false-positive rates.
Precision need
More precise detection methods are needed; traditional cell-based MRD lacks sensitivity.
ctDNA testing has the potential to play an important role across the lymphoma patient journey
Elevating Prognostic Clarity:
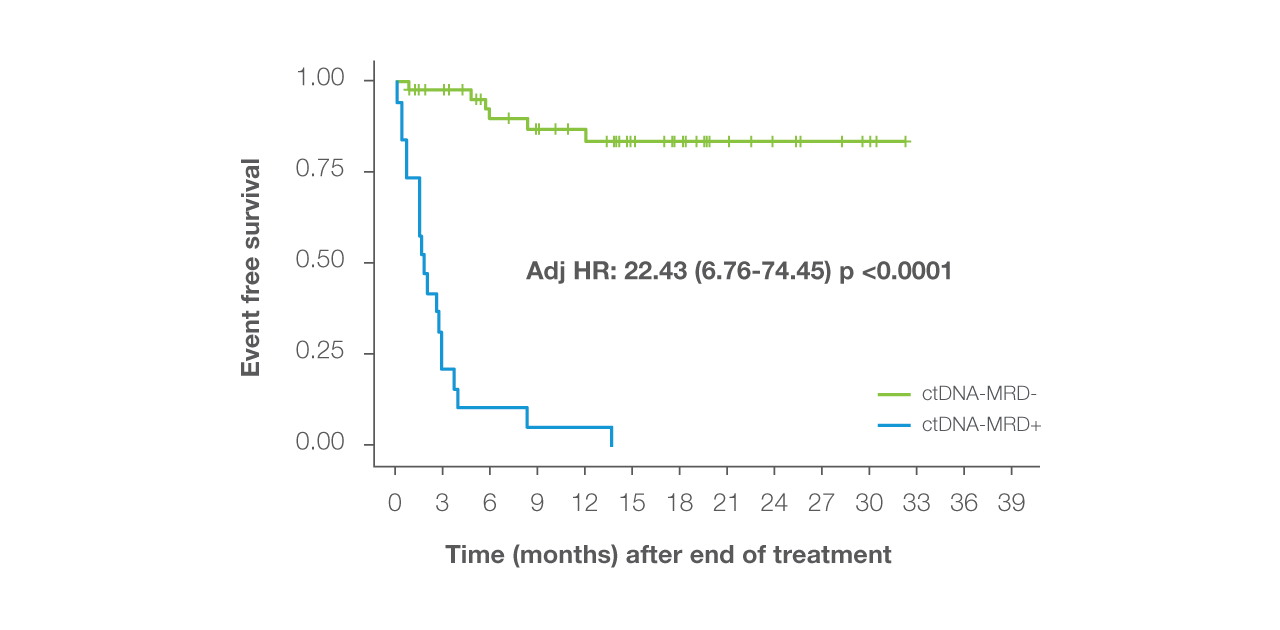
Signatera™ delivers 100% PPV for EFS Event at EOT
Signatera™ achieves 100% positive predictive value (PPV) for EFS event at end of treatment¹
ctDNA-positivity identifies a nearly 50x increased risk¹ of an EFS event
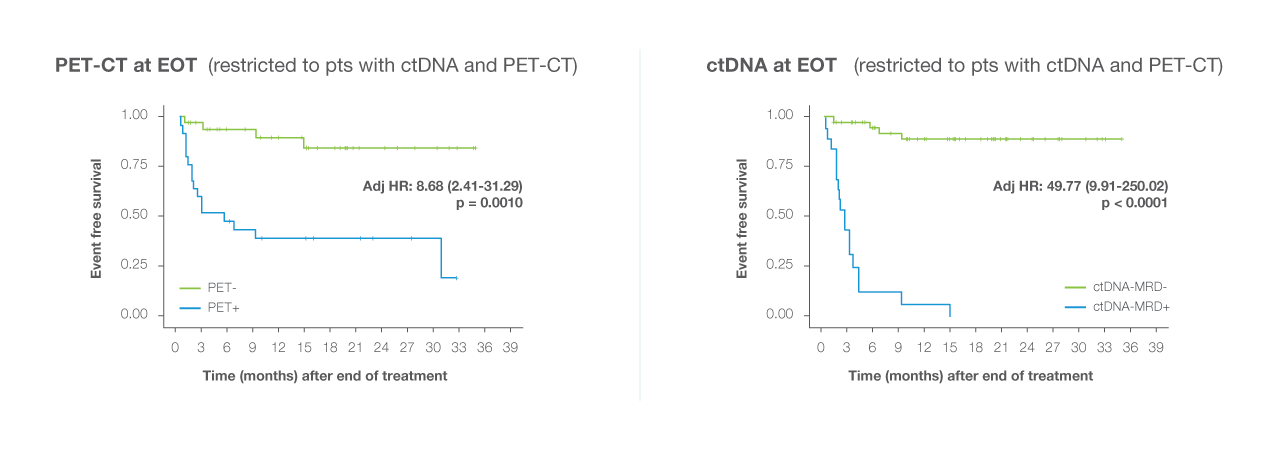
NCCN Guidelines® recommends consideration of ctDNA testing for resolving PET-positive EOT results in DLBCL, when biopsy is not feasible²
Signatera™ achieves 100% positive predictive value (PPV) for EFS event at end of treatment¹
Study design:
This study analyzed prospectively collected, real-world data of MRD detection and ctDNA clearance kinetics in patients with either newly diagnosed or relapsed/refractory lymphoma across 14 subtypes, including a subset of patient who received CAR-T cell therapy.
Enhanced risk stratification
ctDNA-positivity at EOT identifies patients with a nearly 50x increased risk¹ of an EFS event
In a previous real-world study, Signatera™ demonstrated a robust 89% sensitivity and 94% specificity for molecular residual disease detection in patients with DLBCL³

ctDNA-MRD use is now included in the clinical guidelines for DLBCL patients
NCCN clinical guidelines for DLBCL recommend consideration of ctDNA-MRD assessment for adjudicating PET-positive results at the end of treatment, specifically when a biopsy is not feasible²
NCCN = National Comprehensive Cancer Network®. NCCN makes no warranties of any kind whatsoever regarding their content, use, or application and disclaims any responsibility for their application or use in any way.
Building the future of ultrasensitive ctDNA-MRD testing for hematologic malignancies
Natera continues to invest in breakthrough science to push ctDNA-MRD sensitivity beyond today’s limits. Through our December 2025 acquisition of Foresight Diagnostics™, Natera is delivering a step-change in MRD sensitivity. By merging global scale with industry-leading precision, we empower clinicians to track cancer deeper and detect relapse earlier than ever before.

Signatera™ with Phased Variants:
Incorporating phased variants enhances analytical sensitivity and lowers the limit of detection (LOD) for MRD. Launching in 2026, this upgrade builds on the trusted, tumor-informed platform oncologists rely on today.

ctDNA-MRD is a more reliable indicator of relapse risk than imaging alone, particularly when the PET-CT results are unclear or misleading.
A comprehensive oncology portfolio powered by Natera to help guide lymphoma treatment strategies
Natera offers convenient blood draws through the Mobile Phlebotomy service.
Signatera™ MRD test
Tissue sequencing to custom-design each patient’s MRD.
Signatera™ Genome MRD test
For challenging tumor types, genome offers ultrasensitive detection down to 1 PPM.
Altera™ test
Identification of clinically relevant DNA, RNA, and select IHC biomarkers.
Empower™ hereditary cancer test
Uncover clinically relevant genetic mutations that may inform treatment decisions.
In patients with lymphoma, germline testing with Empower™ requires a fibroblast specimen to ensure accurate germline analysis.
Is Signatera™ in lymphoma right for your patients?
We’re here to help you find out
1Galanina N, et al. Real-world evaluation of ctDNA for risk stratification across aggressive and indolent lymphomas. Presented at ASH 2025.
2Referenced with permission from the NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) for B-Cell Lymphomas V.1.2026. © National Comprehensive Cancer Network, Inc. 2026. All rights reserved. Accessed February 19, 2026. To view the most recent and complete version of the guideline, go online to NCCN.org.
3Narkhede M, Tomassetti S, Iqbal M. Tumor-informed ctDNA assessment as a valuable prognostic and predictive biomarker in diffuse large B-cell lymphoma. Frontiers in Oncology. 2024;14.
4Sehn LH, Salles G. Diffuse Large B-Cell Lymphoma. New England Journal of Medicine. 2021;384(9):842-858.
