Two thresholds, one powerful solution
Backed by comprehensive support to make testing easy
Prospera™ is a proven leading indicator of rejection for early detection, timely intervention, and enhanced graft survival. When used for serial monitoring, Prospera™ helps ensure healthy kidney transplants, including for patients with unsuspected, subclinical rejection.
Get a clearer picture of rejection risk with two thresholds
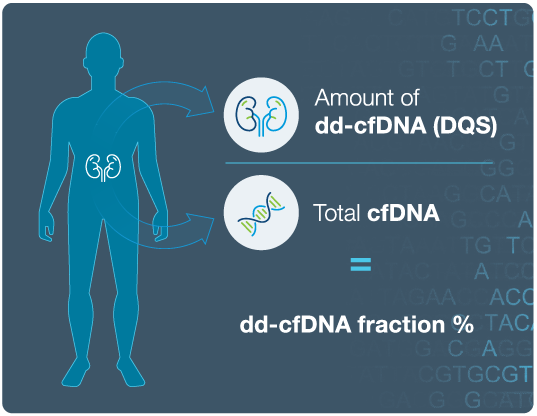
Prospera™ with DQS uses a proprietary technique to estimate both the quantity and the fractions of donor-derived cell-free DNA (dd-cfDNA) in a simple blood test. Prospera™ with DQS is the only dd-cfDNA test to provide two dd-cfDNA related indicators on one report: the percentage of donor-derived cfDNA (dd-cfDNA) and DQS.
Traditional dd-cfDNA tests:
- Detect DNA in the blood from the donor kidney and report this as a fraction of the total cell-free DNA in the blood.
- The fraction of dd-cfDNA may be impacted when the amount of total cell-free DNA is atypically high or low.
DQS is the next evolution of dd-cfDNA results:
- DQS is the estimated quantity of cell-free DNA coming from the donated kidney.
- Unlike traditional dd-cfDNA tests, DQS is independent of fluctuations in levels of total cell-free DNA in the blood.
Benefit from highly sensitive rejection risk with Prospera™ with DQS
The combination of dd-cfDNA fraction and quantity was found to be significantly more accurate than either of these variables alone.1

Discover how Prospera™ with DQS helps inform patient care

Flags otherwise undetected concerns
Patient in mid 50s. At high risk due to COVID-19, BK viral infection, post-transplant donor-specific antibodies (DSA)+, and allograft dysfunction. Nine months post transplant DQS score was highly elevated while the donor fraction estimate was elevated yet far below the threshold for rejection, leading to a biopsy that confirmed TCMR, which may not have happened as quickly otherwise.

Gives insight for faster diagnosis
Patient in mid 70s. At high risk due to high calculated panel reactive antibody (CPRA) with a previous history of biopsy-proven ABMR. Serial testing revealed consecutive results above the DQS threshold, leading to a biopsy that confirmed rejection. Testing with only donor fraction results may not have triggered necessary follow up to find this case of ABMR.

Tests beyond donor fraction
Patient in mid 50s. Pre-transplant patient spent a long time on dialysis and has type II diabetes. Serial monitoring showed a stable donor fraction estimate. The addition of the donor score showed a significant increase at the one year post transplant testing point, which prompted a biopsy that confirmed TCMR. Without the Prospera™ DQS result, this diagnosis may have been missed.
Establish your optimal workflow for easy transition to Prospera™




New: Prospera™ surveillance shows four distinct trends in the immediate post-rejection period
Four distinct dd-cfDNA patterns—categorized as Low, Drop, Mid, and High—helped differentiate those with favorable prognosis from unfavorable prognosis to rejection treatment, providing a non-invasive molecular lens into post-treatment graft health.2
New: Prospera™ now proven to provide accurate prognosis of long-term outcomes following rejection
Results from the first prospective, multi-center trial to examine how donor-derived cell-free (dd-cfDNA) can be used in post-rejection patient management, the PEDAL study, demonstrated that Prospera™ dd-cfDNA trends post-rejection were shown to be strongly associated with and prognostic of one-year outcomes. Prospera™ may help clinicians identify “favorable prognosis” versus “unfavorable prognosis” post rejection treatment, before changes in serum creatinine levels. In this study, creatinine trends had no statistical correlation with outcomes.2
Favorable Prognosis

Low/dropping Prospera™ levels
- 60x more likely to have positive outcomes2
- Suggests these cases mostly responded to treatment
Unfavorable Prognosis

High/elevated Prospera™ levels
- 97.6% (40/41) experience negative outcomes2
- Suggests these cases did NOT respond to treatment
Speak with a Prospera™ expert!
We’re here to help you
References
1Halloran PF, et al. Combining donor-derived cell-free DNA fraction and quantity to detect kidney transplant rejection using molecular diagnoses and histology as confirmation. Transplantation. June 29, 2022. DOI: 10.1097/TP.0000000000004212.
2Bunnapradist,et al. Associations between donor-derived cell-free DNA dynamics and clinical outcomes after kidney allograft rejection: A prospective, multicenter study. American Journal of Transplantation, 2025. doi: 10.1016/j.ajt.2025.07.2470.