New Blood Test Detects Colorectal Cancer Recurrence Up to 16.5 Months Earlier
JAMA Oncology Study Suggests Potential for Natera's Signatera Test to Guide Treatment Decisions After Surgery
SAN CARLOS, Calif., May 9, 2019 /PRNewswire/ — A new clinical study shows that Natera’s Signatera™ test identified colorectal cancer recurrence up to 16.5 months earlier than radiologic imaging by detecting traces of tumor DNA in the blood after surgery. The test also identified patients most likely to relapse, both before and after chemotherapy.1 Results were published in the May issue of JAMA Oncology.
The prospective, multicenter study enrolled 130 patients with stage I–III colorectal cancer from Aarhus University, Randers, and Herning hospitals in Denmark. The study used Natera’s Signatera research-use-only test to evaluate molecular residual disease (MRD) in 829 blood samples collected serially throughout the patient monitoring period.
Results demonstrated that the Signatera test detected molecular recurrence up to 16.5 months earlier than standard-of-care radiologic imaging (average 8.7 months). Serial testing picked up 14 out of 16 relapses (patient-level sensitivity 88 percent), and among patients who did not relapse, 455 out of 456 post-surgical blood samples correctly tested negative (test-level specificity 99.8 percent).
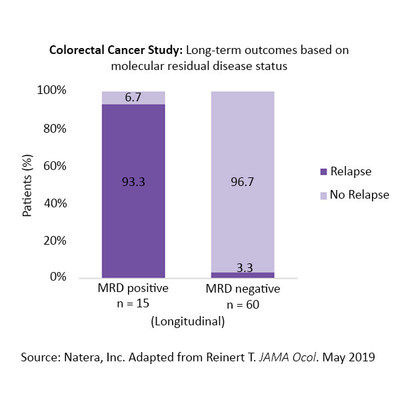
The study also found that MRD status was the most significant predictor of relapse after adjusting for all other known risk factors, including disease stage and lymph node status. Signatera MRD-positive patients who did not receive treatment relapsed in 93 percent of cases. Among patients who remained MRD-negative, the relapse rate was 3 percent. These results underscore the potential of MRD status to risk stratify patients more accurately after surgery to determine which patients need additional therapeutic interventions and which could be safely monitored.
"Our study showed unequivocally that Natera’s personalized multiplex PCR-based next-generation sequencing is a highly sensitive approach for detecting molecular residual disease in the blood," said Claus Lindbjerg Andersen, M.Sc., Ph.D., study lead investigator, Aarhus University. "The results show the potential of blood-based MRD detection to drive a paradigm-shift in how patients are managed during the course of their disease."
"This study highlights Signatera’s potential to change post-operative management of colorectal cancer," said Alexey Aleshin, M.D., MBA, Natera’s oncology medical director. "We look forward to making this test available for clinical practice and pharmaceutical drug trials."
With 1.3 million newly diagnosed cases each year worldwide, colorectal cancer is the third most common cancer and the second leading cause of cancer-related deaths.2 Despite implementation of screening and advances in treatment regimens, the five-year mortality rate remains high at about 40 percent.2-4
The study also reported the first published demonstration of Natera’s plasma-based whole exome sequencing capability, in which there was strong concordance between whole exome results from the plasma and tumor biopsy at time of metastasis.
The study, titled Analysis of Plasma Cell-Free DNA by Ultradeep Sequencing in Patients With Stages I to III Colorectal Cancer, can be found here.
About Signatera™
Signatera is the first custom-built circulating tumor DNA (ctDNA) test for molecular treatment monitoring and molecular residual disease (MRD) assessment. The test is available for research use only (RUO) until its clinical launch planned for Q2 2019. The Signatera methodology differs from currently available liquid biopsy tests, which test for a fixed panel of therapeutically relevant genes. Signatera provides each individual with a customized blood test tailored to match the clonal mutations found in that individual’s tumor tissue. This maximizes accuracy for detecting the presence or absence of MRD in a blood sample, even at levels down to a single mutant molecule in a tube of blood. Signatera also allows researchers to track additional mutations of interest, up to several hundred mutations, for clinical studies.
The body of evidence on the utility of Signatera is growing, with multiple studies demonstrating the Signatera RUO method’s ability to detect molecular residual disease, measure treatment response, and identify recurrence months or years earlier than the standard of care for a variety of cancer types, including breast cancer, early stage non-small cell lung cancer, bladder cancer, and colorectal cancer.1, 5-8 Based on numerous studies across multiple cancer types, a positive Signatera RUO result without further treatment has predicted clinical relapse over 98 percent of the time.1, 5-8
Natera will also offer a research-use-only service for plasma-based whole exome sequencing to create a personalized assay when tissue is not available, or reflexively for Signatera ctDNA positive cases, to characterize resistance mutations, actionable mutations, neoantigens, and tumor evolution. The service will interrogate approximately 20,000 genes from ctDNA to detect somatic mutations, representing a significant increase in coverage over most commercially available fixed liquid biopsy panels. If ordered as a combined service, researchers can first use Signatera to monitor patients for the presence or absence of ctDNA, and for positive patients they can reflex to a plasma exome to characterize tumor evolution using the same exact DNA library sample. Natera expects the service to become available in the second half of 2019.
About Natera
Natera, Inc. (NASDAQ: NTRA) is a global leader in cell-free DNA testing. The mission of the company is to change the management of disease worldwide with a focus on reproductive health, oncology, and organ transplantation. Natera operates an ISO 13485-certified and CAP-accredited laboratory certified under the Clinical Laboratory Improvement Amendments (CLIA) in San Carlos, Calif. It offers a host of proprietary genetic testing services to inform physicians who care for pregnant women, researchers in cancer including biopharmaceutical companies, and genetic laboratories through its cloud-based software platform. For more information, visit Natera.com. Follow Natera on LinkedIn.
Forward-Looking Statements
All statements other than statements of historical facts contained in this press release are forward-looking statements and are not a representation that Natera’s plans, estimates, or expectations will be achieved. These forward-looking statements represent Natera’s expectations as of the date of this press release, and Natera disclaims any obligation to update the forward-looking statements. These forward-looking statements are subject to known and unknown risks and uncertainties that may cause actual results to differ materially, including with respect to our efforts to develop and commercialize new product offerings, our ability to successfully increase demand for and grow revenues for our product offerings, our collaborations with commercial partners such as medical institutions, contract laboratories, laboratory partners, and other third parties, whether the results of clinical studies will support the use of our product offerings, our expectations of the reliability, accuracy and performance of our screening tests, or of the benefits of our screening tests and product offerings to patients, providers and payers. Additional risks and uncertainties are discussed in greater detail in "Risk Factors" in Natera’s recent filings on Forms 10-K and 10-Q and in other filings Natera makes with the SEC from time to time. These documents are available at www.natera.com/investors and www.sec.gov.
This test was developed by Natera, Inc. a laboratory certified under the Clinical Laboratory Improvement Amendments (CLIA). This test has not been cleared or approved by the U.S. Food and Drug Administration (FDA). Although FDA does not currently clear or approve laboratory-developed tests in the U.S., certification of the laboratory is required under CLIA to ensure the quality and validity of the tests.
Contacts
Investor Relations: Mike Brophy, CFO, Natera, Inc., 650-249-9090
Media: Andrea Sampson, Sullivan & Sampson, 714-374-6174, asampson@sullivanpr.com
References
- Reinert T, Henriksen T, Christensen E, et al. Analysis of plasma cell-free DNA by ultradeep sequencing in patients with stages I to III colorectal cancer. JAMA Oncol. 2019.
- Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2015;136(5):E359-E386.
- Iversen LH, Green A, Ingeholm P, Østerlind K, Gögenur I. Improved survival of colorectal cancer in Denmark during 2001-2012 – The efforts of several national initiatives. Acta Oncol. 2016;55 Suppl 2:10-23.
- Osterman E, Glimelius B. Recurrence risk after up-to-date colon cancer staging, surgery, and pathology: Analysis of the entire Swedish population. Dis Colon Rectum. 2018;61(9):1016-1025.
- Christensen E, Birkenkamp-Demtröder K, Sethi H, et al. Early detection of metastatic relapse and monitoring of therapeutic efficacy by ultra-deep sequencing of plasma cell-free DNA in patients with urothelial bladder carcinoma. J Clin Oncol. 2019. DOI:10.1200/JCO.18.02052.
- Coombes RC, Page K, Salari R, et al. Personalized detection of circulating tumor DNA antedates breast cancer metastatic recurrence. Clin Cancer Res. 2019.
- Magbanua M, Brown-Swigart L, Hirst G, et al. Personalized serial circulating tumor DNA (ctDNA) analysis in high-risk early stage breast cancer patients to monitor and predict response to neoadjuvant therapy and outcome in the I-SPY 2 TRIAL. Data presented at spotlight session: San Antonio Breast Cancer Symposium; December 5, 2018. Abstract 1259.
- Abbosh C, Birkbak NJ, Wilson GA, et al. Phylogenetic ctDNA analysis depicts early-stage lung cancer evolution. Nature. 2017; 545(7655):446–451.
SOURCE Natera